Teacher Portal:
Watersheds
Investigation 2 – Lab

ZERO-IN
The italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included supporting the teacher’s understanding of the content being introduced within the CELL.
MINDSET
This Investigation is designed to:
- investigate how the abiotic factors of water velocity, sedimentation, and turbidity affect a watershed,
- investigate how water velocity affects sedimentation and turbidity in a watershed.
BE PREPARED
Teacher Preparation for the Investigation includes the following. This preparation should be done prior to students arriving in the lab.
1. Prepare 200 ml of 50% glycerin solution per class.
a. Measure 100 ml of glycerin in a 250 ml graduated cylinder. Glycerin is very viscous and will stick to the walls of the graduated cylinder. Let it settle to get an accurate reading of the volume.
b. Pour the glycerin into a 400 ml beaker, transferring as much as possible.
c. Add 100 ml of water to the graduated cylinder. Swirl the water gently to rinse any remaining glycerin from the inside of the graduated cylinder.
d. Pour the water into beaker with the glycerin. Stir the solution with a glass stir rod or with a stir bar and stirrer.
e. You may want to prepare enough glycerin for this Investigation and the Performance Assessment at one time. The 50% glycerin solution can be stored in plastic bottles until needed.
2. You may prepare the mixture of 0.5g of flour and 300ml water solution for each student group prior to the Investigation if the anticipated time is limited.
3. Pour 30 ml of the 50% glycerin solution into five 50 ml beakers (one per group). Label each beaker and place the beakers at the distribution center.
4. Turn the spectrophotometers on before class starts to give the equipment time to warm up.
5. Place a box of Kim-wipes, a water bottle with water, one cuvette, and a beaker for waste next to each spectrophotometer.
6. Place the remaining materials at the distribution center.
7. Divide students into five cooperative groups.
Note: Each student lab group will need the materials listed below.
Student Preparation for the Investigation includes having students gather the following materials. This preparation takes place on lab day after student lab groups have settled at their assigned lab tables.
Note: The materials are listed in students’ SDRs. They are also listed below for your reference.
- (1) triple beam balance
- (1) spectrophotometer
- (1) cuvette
- (1) hotplate/stirrer
- (1) stir bar
- (2) weigh dishes
- (1) beaker, 400ml
- (5) centrifuge tubes, 15 ml
- (6) plastic droppers
- (1) wooden test tube rack
- marker
- (1) glass stir rod
- 1.0g flour
- 300ml distilled water
- 30ml 50% glycerin
- (1) stopwatch
- (1) clear metric ruler
- (1) box Kim-wipes
- (1) water bottle with water
- masking tape
INVESTIGATE
-
- Instruct each group to obtain the following materials from the distribution center: one (1) triple beam balance, one (1) stir bar, one (1) hotplate/stirrer, thirty (30) ml 50% glycerin, 1.0g flour, one (1) stopwatch, one (1) lab scoop, one (1) wooden test tube rack, two (2) weigh dishes, one (1) 400 ml beaker, six (6) plastic droppers, five (5) 15 ml centrifuge tubes, masking tape, one (1) clear metric ruler, one (1) glass stir rod, one (1) box Kimwipes, one (1) water bottle with water, one (1) cuvette, one (1) spectrophotometer, one (1) triple beam balance, 300 ml distilled water and one (1) lab marker.
- If students have not had experience using a weigh dish to measure substances on the triple beam balance, demonstrate the procedure to the class. Show students how to account for the mass of the weighing dish by doing a sample calculation on the blackboard. Refer students to the procedures: Triple Beam Balance Use and Operation and Measuring Mass Using a Weigh Dish or Beaker, if necessary.
- When groups are preparing the watershed models, remind students that they will need to place the stir bar in the water before adding the flour.
- Direct students to read the steps before Trial 1 and the steps included in each Trial before they begin the experiment. If necessary, review the directions as a class.
- Caution students that in Trial 1, they should not stir the mixture. As they begin stirring in Trial 2, they may need to use a glass stir rod to move flour away from the stir bar so that it can begin stirring freely on the bottom of the beaker. Once the stir bar begins stirring, students should not shut the stir plate off until Trial 4 is completed.
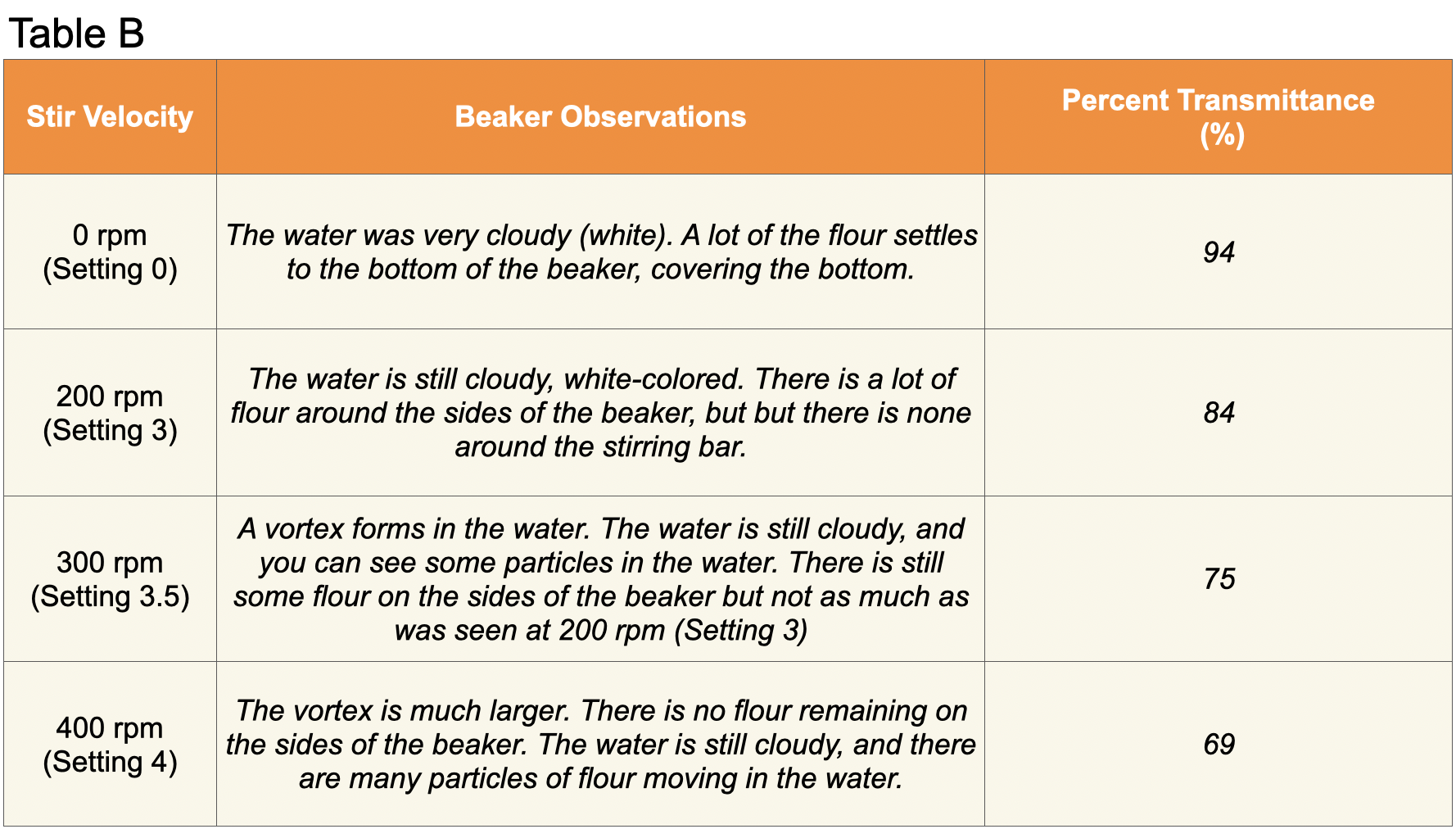
- As students make their observations in Trials 1-4, instruct them to pay close attention to the cloudiness of the water and the sediment along the bottom and walls of the beaker. Students should record or draw their observations in Table B in their Student Data Record.
- Before beginning the next portion of the Investigation, review the use of spectrophotometers with students. Spectrophotometers are laboratory instruments designed to measure the relative amount of light of a particular wavelength that is transmitted or absorbed by a liquid. In this Investigation, students will be looking at the transmittance of light or the amount of light that passes through a substance. When measuring the transmittance of light, the spectrophotometer will report the amount of light transmitted as a percent from 0% to 100%. A value of 100% indicates that all of the light that is released from the light source is transmitted through the liquid. A percentage less than 100% indicates that some light is absorbed by the liquid.
-
- Ask students to read the Background preceding the steps for Trials 5-8 of the Student Data Record. Explain to students that turbidity is the cloudiness of the water. The turbidity of water can be measured analytically through the use of a spectrophotometer. If water is very turbid, less light will pass through the water and it will have a low % transmittance.
- Guide students through the steps of mixing the solution. Students should gently mix the glycerol and sediment solutions before placing them in the spectrophotometer. The glycerol keeps the flour in suspension long enough to get an accurate reading of percent transmittance. Remind students that the “blank” used for the spectrophotometer should be prepared by combining 5 ml of the 50% glycerol solution and 1 ml of water.
- Remind students to set the wavelength of the spectrophotometer to 600 nm.
- Stress to students that to ensure accuracy of their results, they must use the same cuvette for the blank and each sample and place the cuvette in the sample compartment the same way each time. This will account for any variations in the glass. Students should also be careful to avoid getting fingerprints on the cuvettes, wiping with a Kim-wipe before placing the cuvette in the sample compartment.
- If necessary, assist students in graphing water velocity versus percent transmittance and Drawing of a Best Fit Line. Note: May be postponed to PostLab.
CLEAN UP
Let students know your expectations for clean up. Ask them to clean up.

