Teacher Portal:
Weathering and Erosion
Investigation 1 – Lab

ZERO-IN
Italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included to support the teacher’s understanding of the content being introduced within the CELL.
MINDSET
This Investigation is designed to:
- help students recognize that weathering includes both physical and chemical weathering,
- provide students with the opportunity to model physical weathering of marble,
- provide students with the opportunity to model the chemical weathering of marble, and
- allow students to investigate the factors that affect physical and chemical weathering.
BE PREPARED
Teacher Preparation for the Investigation includes the following. This preparation should be done prior to students arriving in the lab.
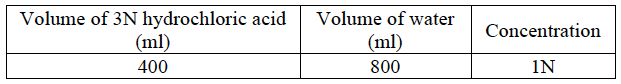
- Prepare 1200 ml of a 1N hydrochloric acid solution using the procedure in the table below.
- For each student group, place ~10g of marble chips in a 50ml beaker. Ten chips approximately 10g.
- For each student group, place ~10g of calcium carbonate in a 50ml beaker. Fill the beaker to approximately 15ml to approximate 10g.
- For each student group, fill a 400ml beaker with 220ml of 1N hydrochloric acid.
- Place all materials at a central location.
- Divide students into five cooperative groups.
Student Preparation for the Investigation includes having students gather the following materials. This preparation takes place on lab day after student groups have settled at their assigned lab tables.
Note: The materials are listed in students’ SDRs. They are also listed below for your reference.
- (1) triple beam balance
- (1) weigh dish
- (1) lab scoop
- (1) 50ml beaker containing marble chips
- (1) 100ml graduated cylinder
- (2) 500ml Erlenmeyer flasks
- (1) 50ml beaker containing calcium carbonate
- (1) 400ml beaker containing hydrochloric acid
- (1) pair of goggles per student
- (1) pair of gloves per student
Direct one student from each lab group to collect the materials listed in their SDRs.
INVESTIGATE
- Inform students that they will perform three Trials during this Investigation.
- Trial 1 introduces students to physical weathering of marble.
- Trial 2 introduces students to chemical weathering of marble.
- Trial 3 introduces students to the combination of physical and chemical weathering.
- Direct students to complete the Recall section in their SDRs.
- Encourage students to reflect on the PreLab video as they move through the procedural steps.
- Explain to students that during the Experiment, every procedural step is important. If one step is skipped, data can become invalid. To help students keep on track, direct them to read each step thoroughly, complete the step, then check it off (Read it – Do it – Check it off).
- Direct students to complete the procedural steps in their SDRs.
Note: The procedural steps are listed below for your reference. Teacher “Notes” are inserted, as needed, to help facilitate the lab.
- In this Trial, you will analyze how physical weathering can affect pieces of marble.
- Using the triple beam balance, lab scoop and a weigh dish, add enough marble chips to obtain a mass of about 5g.
- Remove the weigh dish and place it on the lab table.
- Use the lab scoop and try to physically weather the marble chips by reducing them to smaller pieces.
- Answer the questions.
Trial 2
- In this Trial, you will analyze how chemical weathering can affect pieces of marble.
- Use the 100ml graduated cylinder to measure 50ml of hydrochloric acid and pour it into the 500ml Erlenmeyer flask.
- Add all of the marble chips to the hydrochloric acid in the flask. Swirl the flask so the chips are completely mixed with the acid.
- Observe the chips and answer the questions.
Trial 3
- In Trial 1, you found it difficult to reduce the size of the marble chips because they are too hard.
- In Trial 3, you will use calcium carbonate powder as a model of marble that has been physically weathered and reduced in size to very small pieces. You will analyze how chemical weathering affects these small pieces of marble.
- Using the triple beam balance, weigh dish and lab scoop, add enough calcium carbonate to obtain a mass of 5g.
- Use the 100ml graduated cylinder to measure 50ml of hydrochloric acid and pour it into the second 500ml Erlenmeyer flask.
- Add all of the calcium carbonate powder to the hydrochloric acid in the flask.
- Observe the flask and answer the questions.
CLEAN UP
Let students know your expectations for clean up. Ask them to clean up.
