Teacher Portal:
Solutes and Solubility
Investigation 1 – PostLab

ZERO-IN
Italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included to support the teacher’s understanding of the content being introduced within the CELL.
ANALYZE IT
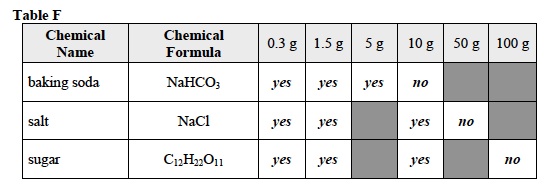
When students have finished their Investigation, use the board to combine the data from each student group and ask students to complete Table F in the Student Data Record.
Note: Questions marked with a triangle (∆) are included to enrich students’ understanding. These questions do not appear in students’ SDRs but should be used as additional discussion points during the PostLab.
Discuss the solubility of each of the compounds in water.
- ∆ Did all masses of each compound dissolve? No, some larger masses of the compounds did not dissolve.
- ∆ Was the mass of each compound that did not dissolve the same or different? The masses for each compound were different.
Instruct students to complete the Analysis Questions in their SDRs then discuss them as a class. Use the suggested responses below to guide students’ answers.
- Compare the solubility of the three compounds in water.

- What was the highest concentration at which baking soda (NaHCO3) was soluble in water? 5 g baking soda per 100 ml water
- What was the highest concentration at which salt (NaCl) was soluble in water? 10 g salt per 100 ml water
- What was the highest concentration at which sugar (C12H22O11) was soluble in water? 50 g sugar per 100 ml water
- Between what masses is the saturation point of each of the compounds?
- baking soda: 5 – 10 g per 100 ml water
- salt: 10 – 50 g per 100 ml water
- sugar: 50 – 100 g per 100 ml water
- Does a solute have a maximum concentration in a certain volume of water? Why or why not? Yes. For each solute, there was an amount of sample that would not dissolve in 100 ml of water. For example 0.3 g, 1.5 g, and 5 g of baking soda dissolved in 100 ml of water, but 10g did not. The maximum concentration of solute that will dissolve in a certain amount of water is the solute’s saturation point. A solute reaches a saturation point because the solvent particles are not able to surround each solute particle.
- Did the different solutes have the same solubility or a different solubility? Each different solute had a different solubility. No more than 5 g of baking soda, 10 g of salt, and 10 g of sugar dissolved in 100 ml of water.
- Look at the chemical formulas for the compounds tested in the experiment. Do compounds composed of the same elements have the same solubility or a different solubility? Compounds composed of the same elements have different solubilities.
GET FOCUSED
Instruct students to complete the Focus Questions in their SDRs then discuss them as a class. Use the suggested responses below to guide students’ answers.
- What affects the solubility of a solute? The unique combination of elements that make up each compound results in the differing solubility of each compound. The different combination of elements determines that each compound is soluble in water up to a different concentration.
- Is there a relationship between the concentration of a solution and the saturation point of a solute? Yes. Solutes are able to dissolve and form solutions only up to concentration that corresponds to their specific saturation points. Any amount of solute greater than the saturation point will not dissolve
