Teacher Portal:
Photosynthesis
Investigation 2 – Lab

ZERO-IN
Italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included to support the teacher’s understanding of the content being introduced within the CELL.
MINDSET
This Investigation is designed to:
- provide students with evidence of the use of carbon dioxide during photosynthesis,
- provide students with evidence of the production of oxygen during photosynthesis, and
- illustrate to students that light is essential in photosynthesis.
BE PREPARED
Teacher Preparation for the Investigation includes the following. This preparation should be done prior to students arriving in the lab.
- For each student group, fill one (1) liter pitcher with 200 ml of bottled or distilled water.
- Fill one (1) liter pitcher with 1000 ml of bottled or distilled water for the groups to share. Use a piece of tape to label this pitcher “Elodea water.”
Note: For best results, the Elodea should be as fresh as possible. It is recommended that it be obtained from the store less than 24 hours before use in this experiment.
Note: Depending on the time of year that this Core Experience is implemented, the Elodea supplied by some pet stores may be less green and healthy than the Elodea purchased at other times. Thus, while photosynthesis still occurs in these plants, the plants may yield less dramatic results than healthier plants. To resolve this issue, add 2.5 g of baking soda to one liter of water. This solution provides Elodea with additional carbon dioxide, aiding in photosynthesis. This water should then be used as the “Elodea water” as students perform the experiment.
- Place all materials at a central location.
Note: Each student lab group will need the materials listed below.
- Divide students into cooperative groups of five students.
Student Preparation for the Investigation includes having students gather the following materials. This preparation takes place on lab day after student lab groups have settled at their assigned lab tables.
Note: The materials are listed in students’ SDRs. They are also listed below for your reference.
- (1) ring stand with clamp.
- (1) liter pitcher with 200 ml water
- (1) stir rod
- (1) oxygen meter
- (1) plastic dropper
- (1) test tube rack
- (2) test tubes
- (2) stopwatches
- (1) metric ruler
- (1) bulb and socket
- (1) scalpel
- (1) 100 ml graduated cylinder
- (1) 100 ml beaker
- Tell one student from each group to fill a 100 ml beaker with 10 ml of phenol red.
- Another student in each group should obtain two (2) stems of Elodea. The student should then fill one (1) 400 ml beaker with 200 ml of water from the pitcher labeled “Elodea water.”
Note: Because these amounts are approximations and do not need to be exact, students do not need to use graduated cylinders to measure volume. They may simply pour the liquids into their beakers and use the graduations on the beakers.
- Direct one student from each lab group to collect the materials listed in their SDRs.
INVESTIGATE
- Remind students that they will continue to investigate photosynthesis by observing carbon dioxide and oxygen quantities in water containing Elodea plants.
- Introduce students to the oxygen meter. Include the following in the introduction:
- The oxygen meter can measure oxygen in air (O2) and oxygen in a liquid (dissolved oxygen).
- Oxygen in the air is measured in percent concentration. Oxygen in a liquid is measured in milligrams per liter.
- On the tip of the oxygen probe is the sensor through which the measurements are taken.

- Encourage students to reflect on the PreLab video as they move through the procedural steps.
- Explain to students that during the Experiment, every procedural step is important. If one step is skipped, data can become invalid. To help students keep on track, direct them to read each step thoroughly, complete the step, then check it off (Read it – Do it – Check it off).
- Direct students to complete the procedural steps in their SDRs
Note: The procedural steps are listed below for your reference. Teacher “Notes” are inserted, as needed, to help facilitate the lab.
Trial 1: Observing the Level of Carbon Dioxide
- Place the stems of Elodea in the beaker of 200 ml of “Elodea water.”
- Use the graduated cylinder to prepare two test tubes of water, each with 25 ml of water.
- Put each test tube in the test tube rack.
- Place 4 drops of phenol red in each test tube and stir the water with the stir rod.
- Hold a piece of white paper behind the test tubes and observe the color of the water.

- Based on its color, do you think that the water is slightly basic (reddish-purple tint) or slightly acidic (orange tint)? The water will likely have a slight orange-red hue rather than a red-purple tint. However, due to differences in water, students’ water may appear slightly acidic or slightly basic.
- Measure a sample of Elodea approximately 10 cm
 in length.
in length. - Place the piece of Elodea in one of the test tubes.
- Move the test tube rack so that the test tubes are 5 cm from the light bulb in the bulb and socket.
- Turn on the light and begin the stopwatch.
- Predict what will occur as you continue to observe the color of the water. Use your knowledge of the raw materials used in photosynthesis and the products of photosynthesis to make your prediction. As photosynthesis occurs, carbon dioxide should be used. As the carbon dioxide in the water in the test tube is consumed, the water should become more basic. A basic solution turns more reddish-purple in phenol red, so the water in the test tube with Elodea should become more purple and less red-orange, while the water in the other test tube should remain red-orange.

- Leave the bulb turned on throughout the investigation, as you will continue to observe changes in the water of the test tubes as changes occur.
- Record: Observe the two test tubes at the times listed in Table A. You may record slightly before or slightly after the listed time if necessary. Sample data is provided. Student answers may vary.


Trial 2: Measuring the Level of Oxygen
- In this Trial, you will test and record the amount of dissolved oxygen in the water in your beaker several times throughout this investigation.
- While you are watching for any color change in the test tubes, equilibrate the oxygen meter by using the procedure Oxygen Meter Calibration.
- As you perform Trial 2, remember to continue with Trial 1. Observe the test tubes at the times listed in Table A and record your observations in Table A.
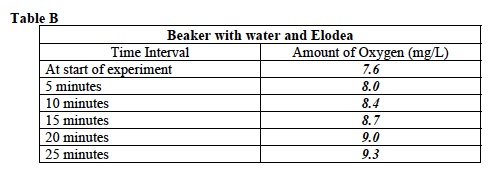
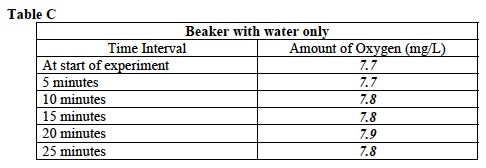
- Record: Some of the class will measure the amount of dissolved oxygen in a beaker containing Elodea. The others will measure the amount of dissolved oxygen in a beaker containing only water. Your teacher will give you your assignment. Circle your investigation below.
beaker with Elodea beaker with water only
Note: If possible, record which students were assigned to each condition. In Investigation Three, students will also investigate two conditions: one involving the effect of light on photosynthesis and one involving the effect of the absence of light on photosynthesis. It would be beneficial if the students running the “No Elodea” control in this investigation were able to run the “Elodea and Light” condition in Investigation Three.
- At the end of the investigation, you will compare your data with the data collected by the other groups for analysis.
- Measure 80 ml of water using a 100 ml graduated cylinder and pour the 80 ml of water into the 100 ml beaker.
- If your group is not using the Elodea, skip step 8.
- If your group is using the Elodea, use a triple beam balance to obtain an Elodea sample with a mass of 5g (approximately 25 cm in length). After you have obtained the sample, cut the Elodea into 1 cm lengths and place them in the beaker.

- Place the beaker 8 cm from the bulb and socket. Move the ring stand so that it is beside the beaker.
- Adjust the clamp on the ring stand so that it is approximately 5 cm from the top of the beaker.
- Place the oxygen probe in the water in the beaker.

A. Tilt the beaker slightly.
B. Hold the oxygen probe so that it enters the water at an angle.
12. Look at the tip of the probe to observe whether or not a large air bubble was formed.
A. If there is a bubble, take the oxygen probe out of the water.
B. Try to insert the probe again.
13. Lower the probe into the water until it is approximately 1 cm from the bottom of the beaker. Make sure the hole for the temperature sensor is covered by the water. Tighten the clamp on the probe until later use.
14. Keep the probe underwater for the entire investigation.
15. Each time you take a reading from the oxygen meter, you will follow these steps. Read all of these steps. Do not begin the procedure until you have read the steps.
A. Make sure the switch on the oxygen meter is on the dissolved oxygen (DO) setting.
B. Gently stir the water in the beaker by rotating  or moving the beaker in a circular motion. Stir for 30 seconds.
or moving the beaker in a circular motion. Stir for 30 seconds.
C. Still stirring, press the record button. The letters “rec” will appear on the meter’s display. Continue to stir the water.
D. Record the oxygen measurements for 30 seconds. Press the recall (CALL) button on the meter 3 times to obtain the average measurement. AVG will show on the screen.
E. Record: Write the average dissolved oxygen concentration in Table B or C.
F. Press the record button again to set up the oxygen meter for the next reading. The letters “rec” will disappear from the meter’s display.
16. Start the stopwatch.
17. Record: Test and record the average amount of dissolved oxygen at the times listed in Tables B and C. Record each measurement of oxygen in the table. Sample data is provided. Student answers may vary.
- As you perform Trial 2, remember to continue with Trial 1. Keep recording your observations in Table A.
CLEAN UP
Let students know your expectations for clean up. Ask them to clean up.
