Teacher Portal:
Light
Investigation 3 – PostLab

ZERO-IN
Italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included to support the teacher’s understanding of the content being introduced within the CELL.
ANALYZE IT
-
Instruct students to complete the Analysis Questions in their SDRs then discuss them as a class. Use the suggested responses below to guide students’ answers.
- How does the absorbance and transmittance of each wavelength of light determine the color of a transparent object? The color of a transparent object is determined by the wavelength that is transmitted through the object. We see that particular color because other wavelengths of light are absorbed by the object. For example, the red solution appears red because the longer orange/red wavelengths of light are transmitted through the solution. Shorter wavelengths of light (green, blue, violet) are absorbed by the solution.
- Graph: Create a separate absorption spectrum for each colored solution (red, green, and blue) to show the relationship between the wavelength of light and the amount of light each solution absorbed. Sample graphs provided.
- Indicate the wavelengths you measured along the x-axis.
- Create a scale of absorbance values along the y-axis.
- Connect the data points.
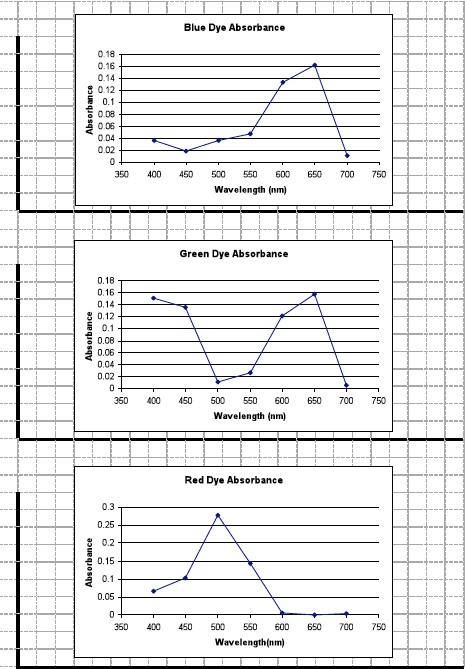
- What pattern do you see between the absorption spectra and the color of the solution? The color of a transparent substance is determined by the wavelength of light transmitted through the substance. We see that particular color or wavelength of light because other wavelengths of light are absorbed by the substance. When we see a color such as red we see that specific color because it absorbs most if not all wavelengths of light except that which corresponds to red wavelengths. The “red” wavelength is transmitted. One general conclusion we can draw concerning colors and how we perceive them is that every color we see is completely different because its absorption spectrum is different from other shades of that color or other colors.
- Why do you think the blue and green spectra both have peaks in the 550-700 nm range while the red spectrum peaks in the 400 to 550 nm range? Refer to the spectrum diagram on page 26 of the Student Data Record if necessary. Blue and green wavelengths are very close to each other in the color spectrum. Therefore, these wavelengths of light behave similarly and absorb at similar wavelengths. Red is at the other end of the color spectrum and thus its behavior is opposite to that of the blue and green wavelengths.
- Why do you think the absorption spectra graph for the green solution has two peaks? The color green absorbs both red and blue wavelengths of light forming two peaks on the graph.
Note: Questions marked with a triangle (∆) are included to enrich students’ understanding. These questions do not appear in students’ SDRs but should be used as additional discussion points during the PostLab.
- ∆ What do you think the transmittance graphs would look like for each solution? Students should indicate that the transmittance graphs for each solution would be in inverse, or opposite, of the absorption graphs. For example, the red wavelength transmittance graph would have its peak around 600 nm – 700 nm and its lowest point around 500 nm. This is because the red solution would absorb the wavelengths between 400 nm and 500 nm and reflect, or transmit, the wavelengths between 600 nm and 700 nm.
Ask students to think back to the first Investigation when they used the spectrophotometer. Remind students that they set the spectrophotometer at 630 nm in order to determine the concentration of the green-colored water.
- ∆ Why do you think the spectrophotometer was set at 630 nm? When determining concentrations of solutions it is common practice to obtain absorbance values at a wavelength that corresponds to the absorption maximum for the solution. The solution tested in Investigation One was green. We see from our experiment today that the absorption maximum for green colored water is 630 nm.
- ∆ If you take a close look at the graph for the green food coloring you’ll notice that it contains two absorption peaks. What conclusions can you draw? The color green absorbs both red and blue wavelengths of light.
- What conclusions can you draw about the colors of opaque objects? Explain your answer. The color of an opaque object results from the reflection of a particular wavelength of light. For example, an object that appears blue reflects wavelengths of light that correspond to the blue end of the spectrum and absorb the longer yellow, orange and red wavelengths of light. However, the pigments used to color objects may reflect more than one wavelength of light. We observed this when viewing several of the gram cubes. One of the blue cubes appeared blue when viewed with the blue filter and green when viewed with the green filter. This suggests that the blue pigment reflected wavelengths of light in both the green and blue areas of the spectrum.

GET FOCUSED
Instruct students to complete the Focus Question in their SDRs then discuss it as a class. Use the suggested response below to guide students’ answers.
- How does wavelength affect the perception of light? The color of a transparent object or medium is determined by the wavelength or wavelengths of light it transmits. We perceive the object or medium as being a particular color because those wavelengths are the ones detected by our eyes. For an opaque object, the color we perceive is determined by the wavelengths of light reflected by the object’s surface. Both transparent and opaque objects absorb all wavelengths of light not transmitted or reflected.
