Teacher Portal:
Kinetic and Potential Energy
Investigation 3 – PostLab
ZERO-IN
Italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included to support the teacher’s understanding of the content being introduced within the CELL.
ANALYZE IT
Instruct students to complete the Analysis Questions in their SDRs then discuss them as a class. Use the suggested responses below to guide students’ answers.
- Think about the chemical reaction of vinegar and baking soda and the information in the Background at the beginning of the Investigation. What caused the circumference of the balloon to change? The circumference of the balloon changed as the balloon began to fill with the carbon dioxide gas formed from the reaction of vinegar and baking soda.
- What type of energy was in the chemical reaction of baking soda and vinegar? Both the baking soda and vinegar had chemical potential energy that was released when the reaction started. Once the reaction began, the potential energy was converted to kinetic energy.
- What type of energy did the balloon have when it was inflating? When it stopped inflating? As the balloon was filling up and moving, it had kinetic energy. Once the balloon stopped moving, it had potential energy because it had moved from its rest position to a higher position.
- Was energy converted during the experiment? Yes. Why? The energy was stored in the bonds of the vinegar and baking soda in the form of chemical potential energy. The chemical reaction released this energy and formed a gas. The energy was transferred to kinetic energy as the balloon blew up. When the balloon stopped moving and was filled with gas, it had potential energy.
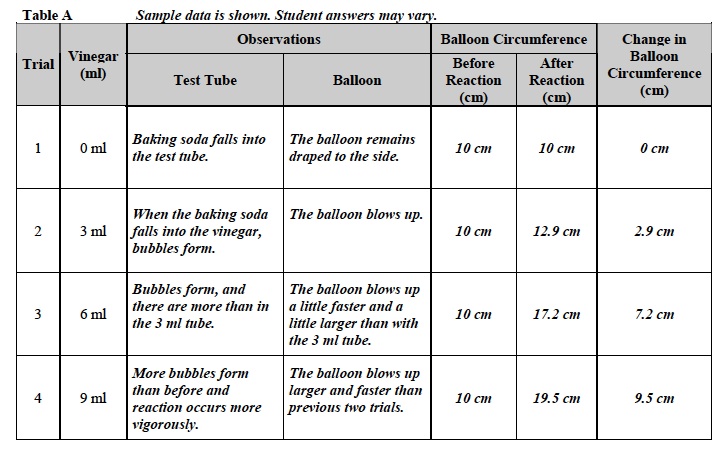
- Look at the observations column of Table A. What signs did you observe that suggest the rate of the chemical reaction and the amount of product formed changed with different amounts of vinegar? The increase in the number of bubbles and the increased speed with which the bubbles appeared were observed as evidence that the rate of the reaction and products of the chemical reaction increased as the amount of vinegar increased.
- How did the amount of chemical potential energy change as the amount of vinegar increased? As the amount of vinegar increased, more vinegar could react with the baking soda and more chemical energy was released. There were a lot more bubbles and the reaction occurred a lot more vigorously (balloon blew up faster and larger) with 9 ml of vinegar than with 3 ml of vinegar.
Note: Students should construct a graph that compares the volume of vinegar in the reaction with the change in circumference of the balloon.
Assist students in graphing and Drawing a Best Fit Line as necessary.
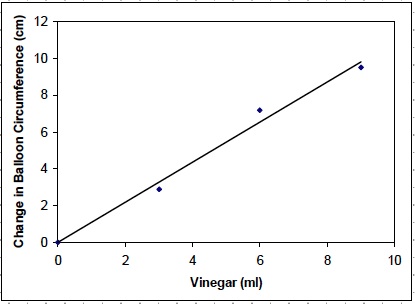
- As the amount of vinegar increased, what happened to the circumference of the balloon? The circumference of the balloon increased as the amount of vinegar increased. With 3 ml of vinegar, the change in circumference of the balloon was 2.9 cm. With 9 ml of vinegar, the change in circumference was 9.5 cm.
- Use the graph to make a prediction. What would the circumference of the balloon be if the volume of vinegar was 5 ml? Use proper units. Student answers will vary based on the data that they obtain. Sample answer based on the above graph: 5.5 cm.
- As the amount of vinegar increased, what happened to the kinetic and potential energy of the balloon? As the amount of vinegar increased, the kinetic and potential energy of the balloon increased. This was observed as the increase in the circumference of the balloon.
- Think about the data in Table A and the graph. Describe the relationship between the amount of vinegar in the reaction, the chemical potential energy of the reaction, and the kinetic and potential energy of the balloon. As the volume of vinegar in the reaction increased, the chemical potential energy of the reaction increased. The increase in chemical potential energy resulted in increases in the kinetic energy and potential energy of the balloon.
Note: Questions marked with a triangle (∆) are included to enrich students’ understanding. These questions do not appear in students’ SDRs but should be used as additional discussion points during the PostLab.
- ∆ Can you think of some ways that the energy from chemical reactions is used in everyday life? Some examples are batteries, inflation of automobile airbags, ice-melting chemicals, light sticks, the light from fireflies (chemiluminescence), chemical cold packs and hot packs, leavening in baking, and campfires (combustion).
GET FOCUSED
Instruct students to complete the Focus Questions in their SDRs then discuss them as a class. Use the suggested responses below to guide students’ answers.
- Can one form of energy be converted to another? Support your answer with data from the experiments. Yes. In this investigation, chemical potential energy was converted to kinetic energy. An increase in chemical potential energy (caused by an increase in the volume of vinegar) showed an increase in both the kinetic energy of the chemical reaction (increase in bubbles and reaction speed) and the kinetic energy of the balloon (change in circumference from 0 cm to 9.5cm).
- How does converting energy from one form to another relate to the Law of Conservation of Energy? The Law of Conservation of Energy says that energy can be converted from one form to another. The direct relationship between the amount of chemical energy (amount of vinegar) and the kinetic energy of the balloon (change in circumference) indicates that energy was conserved. If the Law of Conservation of Energy was not followed, there would not be a direct relationship between the energy conversions.

