Teacher Portal:
Kinetic and Potential Energy
Investigation 3 – Lab

ZERO-IN
Italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included to support the teacher’s understanding of the content being introduced within the CELL.
MINDSET
This Investigation is designed to:
- illustrate the conversion of energy from one form to another,
- introduce students to chemical energy,
- reinforce understanding of the Law of Conservation of Energy as it relates to the conversion of energy, and provide practice in drawing best fit lines and using them to extrapolate data.
BE PREPARED
Teacher Preparation for the Investigation includes the following. This preparation should be done prior to students arriving in the lab.
- Fill five 50 ml beakers (one for each group) to the 25 ml mark with vinegar. Label the beakers “vinegar,” and place them at the distribution center.
- Fill five 50 ml beakers (one for each group) with approximately 3 grams of baking soda. Label the beakers “baking soda,” and place them at the distribution center.
- Use a marker to draw a black line on one side of each uninflated balloon. The line should go across the balloon at the widest point. See Figure 1.
- Place all materials at a central location.
Note: Each student lab group will need the materials listed below.
- Divide students into cooperative groups of five students.
Student Preparation for the Investigation includes having students gather the following materials. This preparation takes place on lab day after student lab groups have settled at their assigned lab tables.
Note: The materials are listed in students’ SDRs. They are also listed below for your reference.
- (1) triple beam balance
- (1) weigh dish
- (1) lab scoop
- (1) test tube rack
- (4) 15 ml falcon tubes
- baking soda
- vinegar
- (1) plastic dropper
- (4) balloons
- (1) clear metric ruler
- (1) 35 cm piece of string
- (1) marker
- masking tape
- (1) calculator
Direct one student from each lab group to collect the materials listed in their SDRs.
INVESTIGATE
- Tell students that a chemical reaction occurs when one or more substances undergo a change to form one or more new substances.
-
- Explain that the new substances are different from the original substances.
- Inform students that some signs of chemical reactions include the formation of gas bubbles, a change in color, and the production of heat.
- Encourage students to reflect on the PreLab video as they move through the procedural steps.
- Explain to students that during the Experiment, every procedural step is important. If one step is skipped, data can become invalid. To help students keep on track, direct them to read each step thoroughly, complete the step, then check it off (Read it – Do it – Check it off).
- Direct students to complete the procedural steps in their SDRs.
Note: The procedural steps are listed below for your reference. Teacher “Notes” are inserted, as needed, to help facilitate the lab.
- Use masking tape to label 4 falcon tubes.
- Label one tube “0 ml vinegar.”
- Label one tube “3ml vinegar.”
- Label one tube “6 ml vinegar.”
- Label one tube “9 ml vinegar.”
- Use a marker to label 4 balloons.
- Label one balloon “0.”
- Label one balloon “3.”
- Label one balloon “6.”
- Label one balloon “9.”
- Place the falcon tubes in the test tube rack. Remove the caps.
- Use a plastic dropper to fill the tubes with vinegar. Do not add any vinegar to the tube labeled “0 ml vinegar.”
- Fill the tube labeled “3 ml vinegar” to the 3 ml mark with vinegar.
- Fill the tube labeled “6 ml vinegar” to the 6 ml mark with vinegar.
- Fill the tube labeled “9 ml vinegar” to the 9 ml mark with vinegar.
- Calculate: Measure the circumference of balloon 0.
- Measure the length of the black line on the balloon.
- Multiply the length by 2 to get the circumference. Circumference = 5 cm × 2 = 10 cm
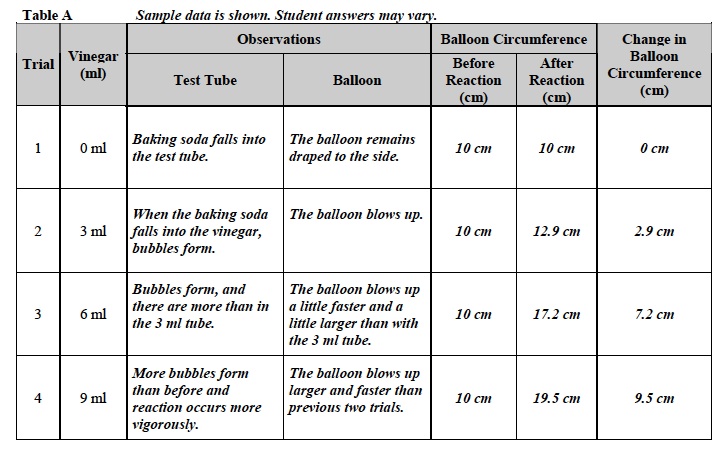
- Record: Write the measurement in Table A under “Balloon Circumference Before Reaction” for 0 ml vinegar.
Note: Table A is on the last page of students’ SDRs.
Trial 1
- In this Trial, you will create the control condition (0 ml vinegar).
- Stretch the balloon.
- Place a weigh dish on the triple beam balance and measure 0.5 g of baking soda according to the Procedure, Triple Beam Balance Use and Operation.
- Gently pour the baking soda into the balloon. Shake the balloon so that the baking soda goes all of the way into the balloon.
Note: Make sure students stretch each balloon before adding the baking soda. This will allow the balloons to inflate more easily in the course of the experiment. Alternatively, students can blow the balloons up slightly and then deflate them.
Note: If students have not had experience using a weigh dish to measure substances on the triple beam balance, demonstrate the procedure to the class. Show students how to account for the mass of the weighing dish by doing a sample calculation on the blackboard. Refer students to the procedure Triple Beam Balance Use and Operation.

- Carefully stretch the neck of the balloon over the top of the falcon tube labeled “0 ml vinegar” so that the balloon hangs to one side. The baking soda should not fall into the falcon tube. The neck of the balloon should be over all of the threads on the top of the falcon tube.

- Lift the balloon so that all of the baking soda falls into the falcon tube. You may need to shake the balloon slightly or tap the tube.
- Let go of the balloon.

- Record: What is happening in the tube? What is happening to the balloon? Write your observations in Table A on the last page of the Investigation 3 Student Data Record.
- Measure the circumference of the balloon using the following steps:
- Do not remove the balloon from the tube.
- Place one end of the string on the balloon.
- Wrap the string around the balloon at the black line.
- Hold on to the string where it meets the end.
- Measure the string from the end to the point you are holding it with the ruler.

- Record: Write the measurement in Table A under “Balloon Circumference After Reaction.”
Trial 2
- Repeat steps 1a through 1f of Trial 1 for the tube labeled “3ml vinegar.”
- Record your observations in Table A.
- Measure the circumference of the balloon. Record the data in Table A.
Trial 3
- Repeat steps 1a through 1f of Trial 1 for the tube labeled “6ml vinegar.”
- Record your observations in Table A.
- Measure the circumference of the balloon. Record the data in Table A.
Trial 4
- Repeat steps 1a through 1f of Trial 1 for the tube labeled “9ml vinegar.”
- Record your observations in Table A.
- Measure the circumference of the balloon. Record the data in Table A.
CLEAN UP
Let students know your expectations for clean up. Ask them to clean up.
