Teacher Portal:
Kinetic and Potential Energy
Investigation 2 – PostLab
ZERO-IN
Italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included to support the teacher’s understanding of the content being introduced within the CELL.
ANALYZE IT
Instruct students to complete the Analysis Questions in their SDRs then discuss them as a class. Use the suggested responses below to guide students’ answers.
- What is the temperature change for each light bulb? To find the change in temperature subtract the starting temperature from the temperature at 6 minutes. Write your answers in the last column of Table C. Sample data is shown below. Student answers may vary.
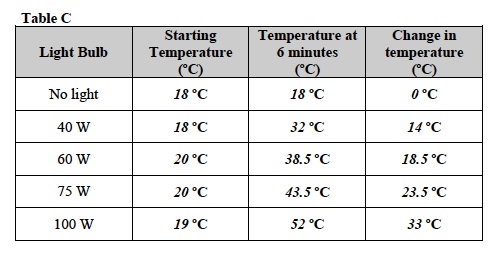
- What form of energy was light converted into in this experiment? Explain your answer. Light energy was converted to heat energy. When the light was not on, there was no heat produced. The temperature did not change. When the light was on, the heat was produced and the temperature increased.
- The temperature of a substance increases when particles of the substance move more quickly. The particles have more thermal energy. Is thermal energy (heat) a type of kinetic or potential energy? Why? Thermal energy is a type of kinetic energy. Kinetic energy is the energy of motion. Since thermal energy is a result of particles moving in a substance, it is kinetic energy.
- Did different light bulbs result in different amounts of thermal energy (heat)? Is there a relationship between the wattage of light bulbs and thermal energy? Support the answer with data from the experiment. Yes. Larger wattage light bulbs produced more heat over 8 minutes than lower wattage bulbs. The 40 W light bulb showed an increase in temperature of 14ºC. The 100 W light bulb showed an increase in temperature of 33ºC .
Note: Students should transfer data from Table B and Table C into Table D. This data will be used to construct a graph of light intensity for each light bulb vs. change in temperature for each light bulb.
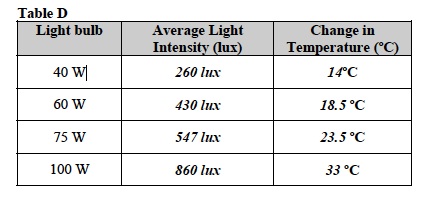 Assist students in graphing and Drawing a Best Fit Line as necessary.
Assist students in graphing and Drawing a Best Fit Line as necessary.
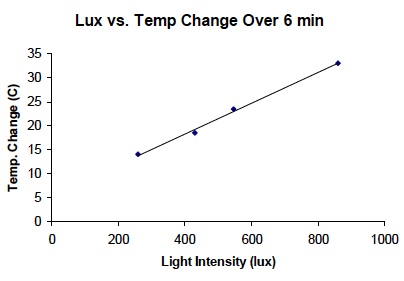
- Look at the graph. How does the amount of light energy of each light bulb relate to the amount of heat energy in the air around each light bulb? Use data to support the answer. As light energy increased the amount of heat increased. Light energy of 260 lux showed an increase in air temperature of 14ºC. A greater amount of light energy (860 lux) showed a greater increase in air temperature (33ºC). There was a direct relationship between light energy and heat energy, as shown by the straight line on the graph.
Analyze the graph as a class.
- Tell students that the straight line on the graph demonstrates a direct relationship between light energy and heat energy.
- This direct relationship indicates that energy was conserved in this system as it was transferred from light energy to heat energy.
- If energy was not conserved, the line would not be straight.
Note: Questions marked with a triangle (∆) are included to enrich students’ understanding. These questions do not appear in students’ SDRs but should be used as additional discussion points during the PostLab.
Review the forms and types of energy observed in Investigation Two with the following questions:
- ∆ What forms of energy were used in this Investigation? Electrical energy, light energy, and heat (thermal energy).
- ∆ Were these forms of energy kinetic energy, potential energy or both? Electrical energy was potential energy until the light was turned on and charges began moving – then it was kinetic energy. Light energy is kinetic energy because light consists of moving photons. Thermal energy is kinetic energy because it is due to the motion of particles in a substance (air in this Investigation).
- ∆ Was energy converted in this experiment? Yes. Energy was converted from electrical energy to light energy to thermal energy (heat). Energy was also converted from potential energy (electrical) to kinetic energy (electrical, light, and heat).
GET FOCUSED
Instruct students to complete the Focus Questions in their SDRs then discuss them as a class. Use the suggested responses below to guide students’ answers.
- Can one form of energy be converted to another? Support your answer with data from the experiments. In this Investigation, students observed the conversion of electrical energy to light energy and the conversion of light energy to thermal energy (heat). Students should support their answers with data from the experiments.
- How does converting energy from one form to another relate to the conservation of energy? Students should recall that the Law of Conservation of Energy states that energy is cannot be created or destroyed but can be converted from one form to another. Both of the graphs in Investigation Two showed direct relationships for the conversion of electrical energy to light and light energy to heat. If the Law of Conservation of Energy was not followed, there would not be a direct relationship in the energy conversions.

