Teacher Portal:
Heat and Heat Transfer
Investigation 3 – PostLab

ZERO-IN
The italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included supporting the teacher’s understanding of the content being introduced within the CELL.
ANALYZE IT
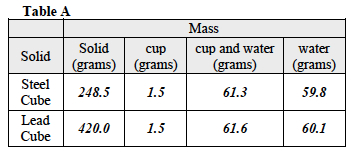
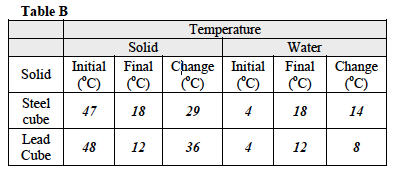
When the students have finished the Investigation, ask them to complete all calculations. Remind them to use the formulas they have been provided to be able to complete their calculations. All data should be recorded in Table A.
Instruct students to complete the Analysis Questions in their SDRs then discuss them as a class. Use the suggested responses below to guide students’ answers.
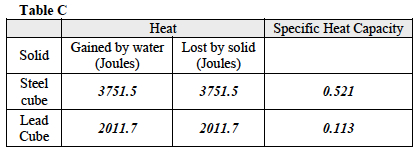
- Which of the two solids has a higher specific heat capacity? The steel cube has a higher specific heat capacity than the lead cube.
- The specific heat capacity of water is 4.184 joules per gram per degree Celsius. How does the specific heat capacity of the two solids compare to the specific heat capacity of water? The water has a higher specific heat capacity than either of the two solids tested.
- Why was a Styrofoam cup used to create the calorimeter? Students should realize that the Styrofoam cup was chosen because it has insulating properties. Therefore, the Styrofoam cup would not have absorbed heat energy from the water, causing a larger change in temperature than would have been correct.
- Would a steel container have worked as well as a Styrofoam cup for calorimetry? Students should realize that the metal is a conductor of heat energy and that the metal container would have allowed heat energy to be absorbed from the water. This would have interfered with the accuracy of determining the specific heat capacity of the two solid substances.
GET FOCUSED
Instruct students to complete the Focus Question in their SDRs then discuss it as a class. Use the suggested response below to guide students’ answers.
- How can the ability to absorb heat be measured as a specific physical property of matter? This Investigation involved calculating a property of matter called specific heat capacity. Specific heat capacity is a measure of the amount of heat energy (joules) required to raise the temperature of 1 gram of a substance by 1 degree Celsius. Different substances require different amounts of energy in order to raise their temperature by 1 degree Celsius, hence they absorb heat energy at different rates. The experiment required the creation of a calorimeter, a piece of equipment designed to prevent heat loss or gain to the substances contained within it. Styrofoam cups were chosen since Styrofoam does not absorb heat energy from the water contained within it, thereby allowing the initial and final temperatures of the water to reflect heat lost from the solid substance alone. Two metals, steel, and lead, were tested. The equation for specific heat capacity was applied and it was determined that steel has a higher specific heat capacity than lead.
COMPREHENSION CHECK
The Comprehension Check is designed to summarize the Core Experience Learning Lab and provide the teacher with an informal way to assess students’ understanding of the big ideas.
If time permits, ask your students to answer each Focus Question below. Use the suggested responses below to guide students’ answers.
- What is the relationship between heat, kinetic energy, and temperature? Heat is the transfer of thermal energy, which includes both kinetic and potential energy. Temperature is a measure of the average kinetic energy of a substance or in an environment. When heat is transferred from areas of higher to lower heat, kinetic energy is transferred from areas of higher to lower kinetic energy (higher to lower temperature). The result is an increase in temperature in the area that once had the lower kinetic energy and a decrease in temperature in the area that once had higher kinetic energy. The transfer of heat stops when the kinetic energy (temperature) of both areas is equivalent.
- What changes in matter accompany changes in heat? In general, matter expands when heated and contracts when cooled because of changes in the kinetic energy of molecules. Changes in heat can also result in changes in the state or phase of matter because of changes in kinetic and potential energy of molecules.
- How do changes in thermal energy relate to the freezing and boiling point of water? The boiling point of water represents the transfer of enough thermal energy to increase the kinetic energy of molecules and break the bonds of molecules so that water changes from a liquid to a gas. The freezing point of water represents the transfer of thermal energy such that the kinetic energy of molecules and the formation of bonds between molecules result in the change of liquid water to solid ice.
- How does the addition of a solute change the freezing and boiling point of water? Adding a solute to water decreases the freezing point of water and increasing its boiling point.
- How can the ability to absorb heat be measured as a specific physical property of matter? This Investigation involved calculating a property of matter called specific heat capacity. Specific heat capacity is a measure of the amount of heat energy (joules) required to raise the temperature of 1 gram of a substance by 1 degree Celsius. Different substances require different amounts of energy in order to raise their temperature by 1 degree Celsius, hence they absorb heat energy at different rates. The experiment required the creation of a calorimeter, a piece of equipment designed to prevent heat loss or gain to the substances contained within it. Styrofoam cups were chosen since Styrofoam does not absorb heat energy from the water contained within it, thereby allowing the initial and final temperatures of the water to reflect heat lost from the solid substance alone. Two metals, steel, and lead, were tested. The equation for specific heat capacity was applied and it was determined that steel has a higher specific heat capacity than lead.
