Teacher Portal:
Heat and Heat Transfer
Investigation 3 – Lab

ZERO-IN
The italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included supporting the teacher’s understanding of the content being introduced within the CELL.
MINDSET
This Investigation is designed to:
- allow students to study heat as a measurable quantity,
- allow students to investigate the conservation of heat across different media, and
- allow students an understand the intrinsic property of specific heat capacity.
BE PREPARED
Teacher Preparation for the Investigation includes the following. This preparation should be done prior to students arriving in the lab.
- Heat 300 ml of water in a 400 ml beaker to 55°C using a hotplate/stirrer. Maintain the water at this temperature by turning the heat control to 1.
- Refrigerate 300 ml of water in a 400 ml beaker overnight. Alternatively, use ice cubes to cool the water to 4°C.
- Place all materials at a central location.
- Divide the class into five cooperative groups.
Student Preparation for the Investigation includes having students gather the following materials. This preparation takes place on lab day after student groups have settled at their assigned lab tables.
Note: The materials are listed in students’ SDRs. They are also listed below for your reference.
- (1) “hot hands” protector
- (3) Styrofoam cups
- (1) steel cube
- (1) lead cube
- (1) stopwatch
- (1) pair of tongs
- (1) triple beam balance
- (1) 400 ml beaker containing 200 ml of 60°C water
- (1) 400 ml beaker containing 200 ml of 4°C water
- (2) 400 ml beakers
- (1) 100 ml graduated cylinder
- (1) thermometer
- (1) calculator
Direct one student from each lab group to collect the materials listed in their SDRs.
INVESTIGATE
- Inform students that in Trial 1, they will determine the specific heat capacity of a steel cube.
- Inform students that in In Trial 2, they will determine the specific heat capacity of a lead cube.
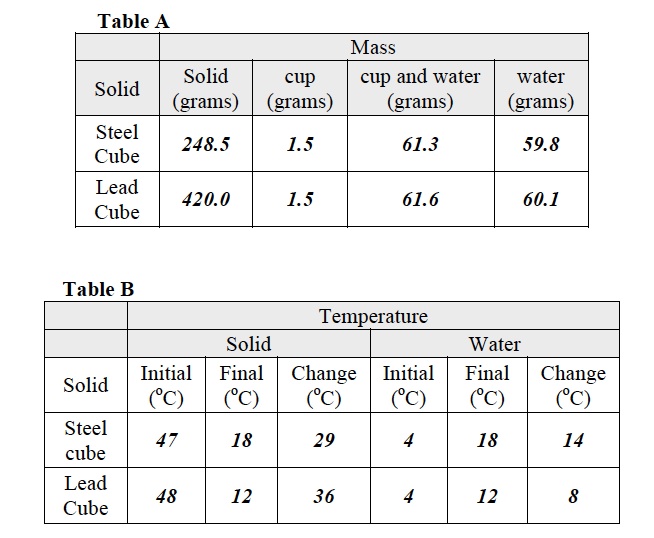
- Inform students that all data from Trial 1 and 2 should be recorded in Tabled A at the end of the Investigation.
- Explain to students that during the Experiment, every procedural step is important. If one step is skipped, data can become invalid. To help students keep on track, direct them to read each step thoroughly, complete the step, then check it off (Read it – Do it – Check it off).
- Encourage students to reflect on the PreLab video as they move through the procedural steps.
- Direct students to complete procedural steps in their SDRs.
Note: The procedural steps are listed below for your reference. Teacher “Notes” are inserted to help facilitate the lab.
Trial 1
- Use a graduated cylinder to add 200 ml of 55oC water to a 400 ml beaker.
- Record the mass of the steel cube in Table A at the end of the Student Data Record.
- Use tongs to transfer the steel cube to the beaker of hot water.
- Create a calorimeter by completing the following steps.
- Determine the mass of one of the two Styrofoam cups. Record the mass in Table A.
- Place one cup in an empty 400 ml beaker. Pour 60 ml of 4oC water into the second cup. Find the mass of the cup and its contents. Record this in Table A.
- Place the water-filled cup inside the first cup in the 400 ml beaker.
- Calculate the mass of the water by subtracting the mass of the cup from the mass obtained in step 4. Record the mass of the water in Table A.
- Record the temperature of the 4oC water in the Styrofoam cup in Table B.
- Record the temperature of the hot water in Table B.
- Use tongs to carefully pick up the steel cube. Place the steel cube into the Styrofoam cup filled with cold water.
- Immediately start the stopwatch at the time zero.
- Measure the temperature of the water, and then at 1 minute intervals until the water temperature stops increasing.
- Record the final temperature of the water in both columns in Table B.
Trial 2
- Use the triple beam balance to weigh the lead cube. Record the mass in Table A.
- You may use the mass of the cup that you determined in step 4.
- Repeat the steps from Trial 1 using the lead cube instead of the steel cube.
CLEAN UP
Let students know your expectations for clean up. Ask them to clean up.
