Teacher Portal:
Heat and Heat Transfer
Investigation 2 – PostLab
 ZERO-IN
ZERO-IN
The italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included supporting the teacher’s understanding of the content being introduced within the CELL.
ANALYZE IT
When students have finished, direct them to Table A and discuss the data.
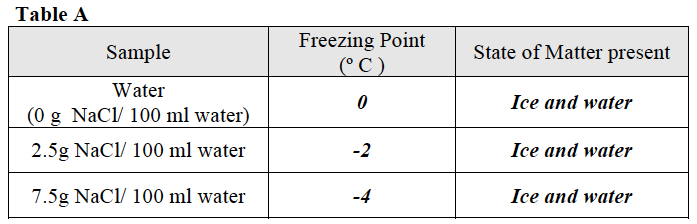
Ask students to use the data in Table A to create a graph that shows the relationship between the amount of salt in water and the freezing point of water.
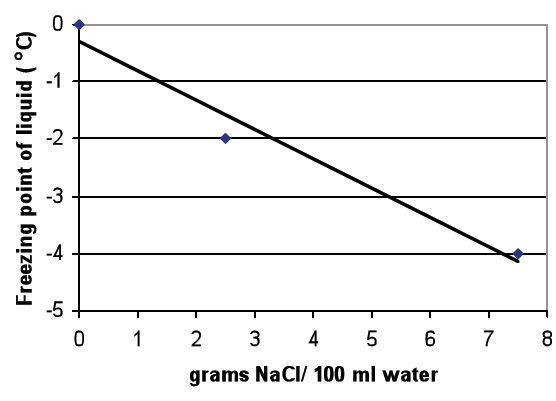
Once students have completed the graph, direct them to use the graph and their observations from Trial 1 to answer the questions following the graph.
- What happened to the freezing point of the water as the amount of NaCl in the water increased? As the amount of NaCl in the water increased, the freezing point of the water decreased.
- Make a prediction. What would the freezing point of water be if the concentration of NaCl was 5g/ 100 ml water? Approximately -3.2o C.
- Would there be a difference in the freezing point of water in a freshwater lake compared to the ocean? Explain why or why not. Water in a freshwater lake has a lower concentration of salt (NaCl) than the water in an ocean. Students should indicate that based on the difference in salt concentration, they would expect a difference in the freezing point of water in a freshwater lake and the ocean. The increased concentration of salt in ocean water would result in a lower freezing point. In other words, students should predict that the water in a freshwater lake would freeze before the water in an ocean.
- Which states of matter were present when pure water reached its freezing point? Student answers may vary depending upon their sample. Students should indicate that either a mixture of ice and water or all ice was present in the test tube when the water reached its freezing point.
- Which states of matter were present when the saltwater reached its freezing point? Student answers may vary depending upon their sample. Students should indicate that either a mixture of ice and water or all ice was present in the test tube when the water reached its freezing point.
- Did the addition of salt change a physical or chemical property of water? Adding salt changed the freezing point of water. The freezing point of water is a physical property of water. The chemical composition of water did not change, only the temperature at which liquid water changed into solid ice.
- Why do you think salt is added to the road when it is snowing or icy? Students should suggest that salt is added to decrease the freezing point of water. Another way of stating this would be to indicate that salt would melt the ice because the freezing point of water had been lowered.
- Why do you think you used a saltwater ice bath in Investigations 1 and 2 when performing experiments in which it was necessary to freeze water? Student answers may vary. Students should suggest that using an ice bath in which salt was added would decrease the freezing point of water. That is, it would allow the temperature of the ice bath to drop below 0oC. As a result, it would create an environment that was cold enough for pure water and water with a small concentration of salt to freeze. If a pure water ice bath were used, the temperature would only be able to reach 0oC. However, because of heat transfer from the surrounding, the temperature might not have remained above 0oC. Using the saltwater ice bath was a way to ensure a temperature below 0oC.
When students have finished, direct them to Table B and discuss the data.
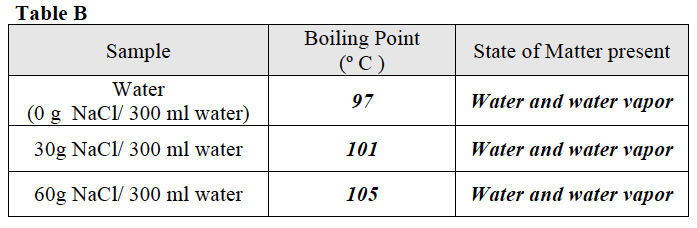
Ask students to use the data in Table B to create a graph that shows the relationship between the amount of salt in water and the freezing point of water
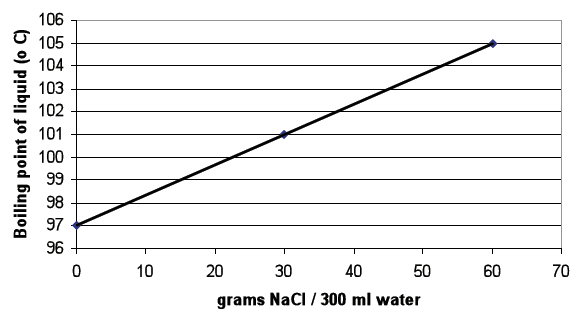
Once students have completed the graph, direct them to use the graph and their observations from Trial 1 to answer the questions following the graph.
- What happened to the boiling point of water as the amount of salt in the water increased? The boiling point of water increased. Water with salt added boiled at a temperature higher than pure water.
- What would you predict the boiling point of water would be if 45 g of salt were dissolved in 300 ml water? 103oC
- Cooks often add salt to the water when boiling pasta. They claim that it makes the water “hotter.” Is this true? Explain your answer. Students should indicate that this is true. Based on data from their experiments they observed an increase in the temperature at which water boiled when salt was added to the water. Therefore, the water with salt added was “hotter” than pure water.
- Which states of matter were present when pure water reached its boiling point? Liquid water and water vapor were present. Water vapor was released from the boiling water into the atmosphere.
- Which states of matter were present when the saltwater reached its boiling point? Liquid water and water vapor were present. Water vapor was released from the boiling water into the atmosphere.
- Did the addition of salt change a physical or chemical property of water? Adding salt changed the boiling point of water. The boiling point of water is a physical property of water. The chemical composition of water did not change, only the temperature at which liquid water changed into water vapor (gas).
Read the Background box in the Analysis Section and then answer the following question as a class.
- Why does the addition of ethylene glycol to water make antifreeze a good solvent for engines in BOTH cold and hot temperatures (winter and summer)? Students should indicate that the addition of ethylene glycol to the water would result in freezing point depression and boiling point elevation of the water. This would mean that the antifreeze would freeze at a temperature lower than 0oC and boil at a temperature higher than the boiling point of pure water at that altitude and barometric pressure. Therefore the solvent running through engines is less likely to freeze in the winter and boil over in the summer.
GET FOCUSED
- How do changes in thermal energy relate to the freezing and boiling point of water? The boiling point of water represents the transfer of enough thermal energy to increase the kinetic energy of molecules and break the bonds of molecules so that water changes from a liquid to a gas. The freezing point of water represents the transfer of thermal energy such that the kinetic energy of molecules and the formation of bonds between molecules results in the change of liquid water to solid ice.
- How does the addition of a solute change the freezing and boiling point of water? Adding a solute to water decreases the freezing point of water and increasing its boiling point.
