Teacher Portal
Chemical Reactions
Investigation 3 – PreLab
ZERO-IN
Italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included to support the teacher’s understanding of the content being introduced within the CELL.
ASK WHY
Remind students that everything in the environment, whether naturally occurring or man-made, is composed of chemicals, and research of chemicals has led to the discovery and development of new and improved paints, medicines, cosmetics, electronic components, and thousands of other products.
BRANCH OUT
Remind students that pharmacists distribute prescription drugs to people. To do this safely, they must understand the chemical composition of these drugs so that they can create special versions of medications for patients who may have allergies to certain substances, give the right dosage, and give patients advice on the possible side effects.
GET FOCUSED
Inform students that the Investigation is designed to help them to answer the following Focus Question:
- In a chemical reaction, what is the relationship between the amount of reactants and the amount of products? As the amounts of the reactants increase, the amounts of the products produced also increase.
Note: This question is located in students’ SDRs at the beginning and end of the Investigation.
Note: This is a succinct response to the Focus Question and is placed here for your reference at this time. A fully developed response to the Focus Question can be found on the PostLab page.
GO DEEPER
As a class, read the Background(s) in the Investigation. Have students read the information aloud or silently to themselves. When students have finished, discuss the following concepts as a class:
- In a chemical reaction, the reactants interact to form the products.
- The consumption of the reactants takes place at the same rate as the production of products.
- To measure the rate of a reaction, scientists measure the disappearance of one of the reactants or the production of one of the products.
- A catalyst affects the rate of a reaction.
- A catalyst is neither a reactant nor a product since it is not consumed or produced in a chemical reaction.
Note: These concepts are integrated into the Background(s) and are used to deepen students’ comprehension of the big ideas.
LEARN THE LabLearner LINGO
The following list includes Key Terms that are introduced in the Investigation Background(s). They should be used, as appropriate, by teachers and students during everyday classroom discourse.
- rate
- wavelength
- nanometer
- spectrophotometer
- absorbance
- catalyst
Note: Definitions to these terms can be found on the Introduction page to the CELL.
Note: Additional words may be bolded within the Background(s). These words are not Key Terms and are strictly emphasized for exposure at this time.
SET FOR SUCCESS
- Inform students that the following chemical equation describes the reaction that they will analyze in this Investigation. To aid students’ understanding, write the equation on the board.
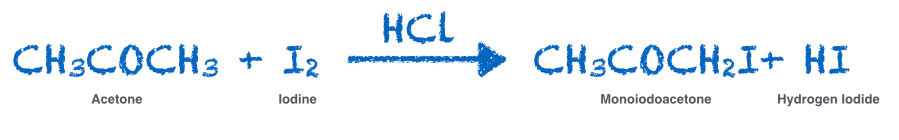
- Explain to students that acetone and iodine are the two reactants and that monoiodoacetone and hydrogen iodide are the two products of the chemical reaction.
- Explain to students that the HCl is a catalyst for the reaction and is neither a reactant nor a product. It does not participate in the reaction directly but serves to speed up the reaction. Since the same amount of HCl is added to the reaction in each Trial, its effect on the rate of the reaction in each Trial will be the same.
- Assess students’ prior knowledge and their understanding of the Background by asking them to predict the answers to the following questions.
- Do chemical reactions always produce products at the same rate?
- What factors affect how fast a chemical reaction takes place?
- If you change the amount of a reactant, what happens to the time it takes to complete a chemical reaction?
Note: Students will formulate answers to these questions after they have completed the Investigation.
- Play the video below. Stop to ask students questions or answer students’ questions when necessary. Remind students to follow along with their SDRs and make any notes that they think might be helpful.
- After the video, direct students to divide into their lab groups to discuss their strategy for the lab. For example, they may assign certain group members to perform specific functions during the lab.
Note: The purpose of the video is to allow students to anticipate the laboratory experience they will soon encounter. Students should leave this PreLab session with a firm idea of what to expect and how to perform in the lab.
Note: Homework is posted below the video.
HOMEWORK
Tell students that they should review the Investigation in preparation for the Lab.

