Teacher Portal:
Chemical Reactions
Investigation 3 – PostLab

ZERO-IN
Italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included to support the teacher’s understanding of the content being introduced within the CELL.
ANALYZE IT
When students have finished their Investigation, direct their attention to their data in Table B.
For each Trial, instruct students to convert the reaction time as it is recorded in Table B from minutes and seconds to seconds. The reaction times in seconds should be entered in the last column of Table B.
Note: A sample calculation is shown in step 1 of the Analysis section in their Student Data Record.
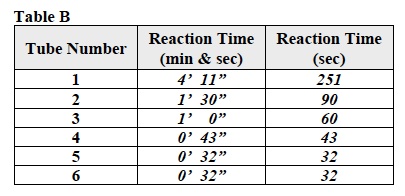
Based on the data in Table B, discuss the questions in problems 3-8 of the Analysis section in students’ Student Data Record. Students should refer to the equation in their Student Data Record.
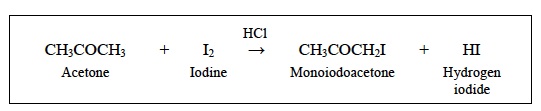
- Which reactant was added using a different volume for each reaction? Acetone (CH3COCH3) was added.
- Did the volume of that reactant increase or decrease for reactions 1 to 6? The volume of reactant increased.
- Which chemical was the other reactant in the reaction? Iodine (I2) was the other reactant in the reaction.
- Was the other reactant added using a different volume for each reaction? No, the same amount of reactant was added for each Trial.
- Which chemical was the catalyst in the reaction? The catalyst was hydrochloric acid (HCl).
- Was the catalyst added using a different volume for each reaction? No, in each Trial the same volume of catalyst was added.
Based on the data in Table B, discuss the questions in problems 9-12 in students’ Student Data Record. Students should refer to the equation in their Student Data Record.
- Reaction time was measured by following the disappearance of which reactant? Iodine
- What evidence supports the idea that the iodine (I2) was consumed in the reaction? The color of the reaction just after the iodine was added was reddish brown because of the iodine. At the end of the reaction, it was colorless suggesting that the iodine had been consumed in the reaction.
- How was the disappearance of the iodine (I2) measured? The disappearance was measured by measuring the color change in the reaction. This was done by measuring the absorbance of the reaction at 460 nanometers using the spectrophotometer.
Ask students to answer the Investigation by completing the following question.
- What was the purpose of this Investigation? Student answers will vary. The purpose of the investigation was to investigate what effect increasing the amount of one reactant had on the reaction time.
Ask students to graph their data from Table B.
Note: Students should connect the data points with a series of straight lines and not draw a best fit line. If necessary, refer students to the procedure for the Graphing of Independent and Dependent Variables.
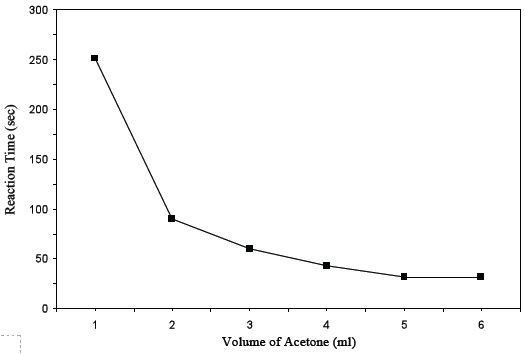
Based on their graph of the data, ask students to discuss the questions in problems 14-17 about the change in reaction time.
- Why was the graph created using the different volumes of acetone and the reaction time? Student answers will vary. The graph was created using the different volumes for acetone and the reaction rate since the acetone (CH3COCH3) was the independent variable and the reaction rate was the dependent variable. The graph illustrates how the change in the amount of one reactant, acetone, affects the reaction time.
- Did the reaction time for Trials 1 to 6 increase or decrease? The reaction time decreased.
- Which reactant caused the reaction time to change? Acetone (CH3COCH3)
- How would you describe the relationship between the amount of the acetone reactant and the reaction time? As the volume of acetone increased, the reaction time decreased.
Discuss students’ answers to the questions in problems 18 and 19 in their Student Data Record. Ask students to refer to the equation.
- As the products were made what happened to the iodine? Student answers will vary. As the products were made, the iodine was consumed.
- What happened to the products as the amount of the reactant, acetone, was increased? Student answers will vary. The rate of production of the products increased as the amount of acetone (CH3COCH3) increased.
Based on the data collected in the Investigation, ask students to answer the following questions, and compare their conclusions with their predicted answers.
- Do chemical reactions always produce products at the same rate? No, the rates of chemical reactions can be different.
- What factors affect how fast a chemical reaction takes place? The amount of a reactant can affect how fast a chemical reaction takes place.
- If you change the amount of a reactant, what happens to the time it takes to complete a chemical reaction? Adding more of one of the reactants compared to the other reactants results in a faster reaction time (decreased reaction time).
GET FOCUSED
Instruct students to complete the Focus Question in their SDRs then discuss it as a class. Use the suggested response below to guide students’ answers.
- In a chemical reaction, what is the relationship among the reactants, the products, and the time it takes to complete the reaction? As the amount of a reactant increases, the reaction time decreases. As the time decreases, this can be measured as an increase in the rate of disappearance of the reactants and an increase in the rate of production of the products. Therefore the more reactants added to a reaction, the more products that will be produced, and the faster they will be produced.
COMPREHENSION CHECK
The Comprehension Check is designed to summarize the Core Experience Learning Lab and provide the teacher with an informal way to assess students’ understanding of the big ideas.
If time permits, ask your students to answer each Focus Question below. Use the suggested responses below to guide students’ answers.
- In a chemical reaction, what is the relationship between the amount of reactants and the amount of products? As the amounts of the reactants increase, the amounts of the products produced also increase. In Investigation One, as the amounts of the NH4OH and the HCl increased, the amount of heat produced increased.
- In a chemical reaction, do the amounts of the reactants affect the amounts of products produced? Reactants must be present in the reaction in equivalent amounts to produce the maximum amount of all reactants. In Investigation Two, the HCl was present in a greater amount than the magnesium since all the magnesium was consumed by the reaction to produce H2 gas. When the amount of magnesium was increased from 0.5 to 1 gram, the excess HCl was able to react and produce more H2.
- How does the Law of Conservation of Matter relate to chemical reactions? No matter is lost from a chemical reaction even if the reactants are not present in equivalent amounts. Every atom of every reactant can be found in either the products or in reactants that are not consumed. In Investigation Two, the atoms of the HCl were found either in the unreacted HCl or in the H2 produced. In every reaction of the Investigation, all of the magnesium atoms were converted to the product, MgCl2.
- In a chemical reaction, what is the relationship among the reactants, the products, and the time to complete the reaction? As the amount of a reactant increases, the reaction decreases. The change in reaction time can be measured either as an increase in the rate of disappearance of a reactant or an increase in the rate of production of a product. Therefore, the more reactants added to a reaction, the more quickly the products will be produced. In Investigation Three, as the concentration of the reactant acetone increased, the reaction time decreased (reaction occurred more quickly) as evidenced by the more rapid disappearance of the colored reactant, I2.
