Teacher Portal:
Chemical Reactions
Investigation 1 – PostLab

ZERO-IN
Italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included to support the teacher’s understanding of the content being introduced within the CELL.
ANALYZE IT
-
When students have finished their Investigation, discuss the data in Table A.
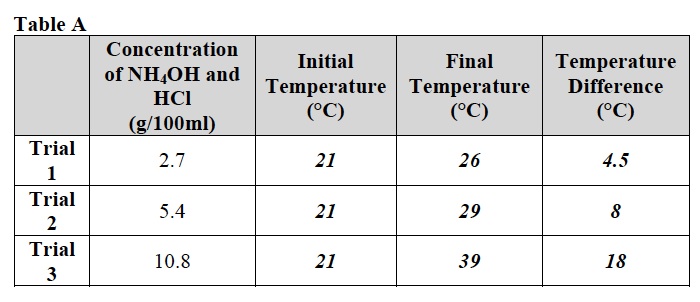
Ask students to complete the analysis of their data by drawing a best fit line in problem 1d of the Analysis section in their Student Data Record.
Note: If necessary, students should refer to the procedures for Determination and Graphing of Independent and Dependent Variables Drawing of a Best Fit Line and Graphing of Independent and Dependent Variables
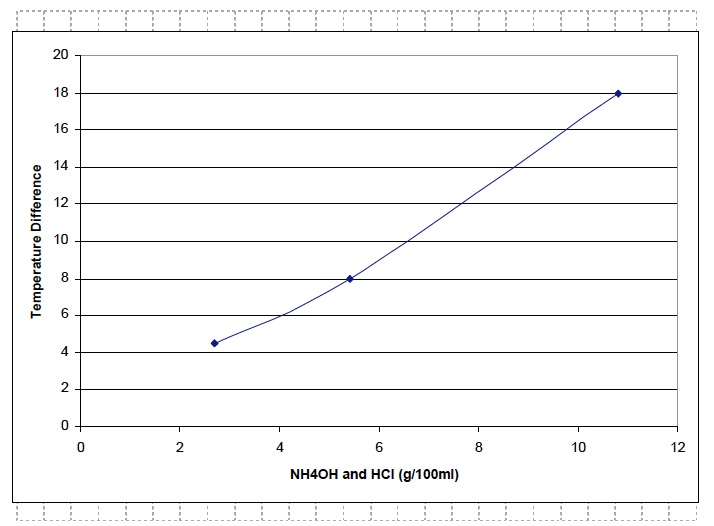
Ask students to analyze their graphs by answering the question. Use the suggested response below to guide students’ answers.
- How does the concentration of the reactants affect the amount of heat produced by the reaction? As the concentration of ammonium hydroxide (NH4OH) and hydrochloric acid (HCl) increased from 2.7 g/100ml to 5.4 g/100 ml to 10.8 g/100 ml, the temperature difference between the initial and final temperatures of the reaction increased from 4.5° C to 8° C to 18° C. Therefore as the amount of each reactant increased, the amount of heat produced also increased.
Ask students to think about the signs of a chemical change and answer the following question. Use the suggested response below to guide students’ answers.
- Since the appearance of the liquids in the reaction did not change after both reactants were added to the cup, what other sign suggested that a chemical change occurred? The release of heat is one of the common signs of a chemical change. In each of the Trials, there was an increase in the temperature of the liquid after both reactants were combined. The increase in temperature suggested that heat was released as a result of a chemical change or chemical reaction.
Based on the data collected in the Investigation, ask students to answer the Conclusion questions then compare their conclusions with their predicted answers. Use the suggested responses below to guide students’ answers.
- What happens to the reacting chemicals in a chemical reaction? The reacting chemicals were used up and converted to the products.
- What is produced in a chemical reaction? The products water, ammonium chloride, and heat were produced.
- How do you know that a chemical reaction has occurred? The release of heat is a common sign of a chemical change.
GET FOCUSED
Instruct students to complete the Focus Question in their SDRs then discuss it as a class. Use the suggested response below to guide students’ answers.
- In a chemical reaction, what is the relationship between the amount of reactants and the amount of products? As the amounts of the reactants increase, the amounts of the products produced also increase.
