Teacher Portal:
Atomic Structure
Investigation 3 – Concept Day
ZERO-IN
Italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included to support the teacher’s understanding of the content being introduced within the CELL.
ASK WHY
Remind students that it is important to know about atoms because atoms are the building blocks of matter. We use our knowledge of atoms to explain the properties of matter, understand and manipulate matter and, ultimately, the world around us.
BRANCH OUT
Explain to students that nuclear engineers use the energy in the bonds of atoms to create electricity for us. They do this through a process called fission wherein uranium atoms are split. The thermal energy produced from splitting the uranium atoms turns water into steam, which in turn drives turbine generators to produce electricity.
PRINT IT
Use your browser to download a printable PDF as a help during the slide presentation and to make additional notes. In your browser, go to File > Print and then choose to save as PDF.
NAVIGATE IT
Once the slide presentation is launched
- use your left and right arrows to advance or go back in the slide presentation, and
- hover your mouse over the left edge of the presentation to get a view of the thumbnails for all the slides so that you can quickly move anywhere in the presentation.
- Click HERE to launch the slide presentation for the CELL.
SHARE IT

SLIDE ATOM-3-1
- The Lab for this Investigation involves using yet another physical property of elements, electrical conductivity, as a means of further exploring the placement of elements on the Periodic Table of the Elements.
- The process of conducting an electric current, a property of what are called conductors, involves charges of subatomic particles in certain atoms.
- We will therefore also take this opportunity to build upon our previous discussion of subatomic particles to include the charges of protons, neutrons, and electrons.
_______________________________________

SLIDE ATOM-3-2
- Note: This slide shows the Periodic Table of the Elements. It is included simply to refresh students’ memory of the Table and to illustrate the position of the element copper (Cu), which we will explore in more detail in the next slide.
Note: The Periodic Table may also be used by the teacher to review the overall layout of the Table or to encourage further practice in reading it.
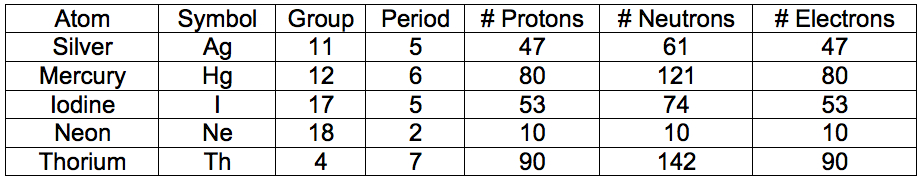
Note: The table below is presented as a guide for teachers who wish to challenge their students with additional practice while this slide is up:
_______________________________________

SLIDE ATOM-3-3
-
Note: This slide depicts the copper atom.
- Prior to clicking the ANIMATION, have students look at the information from the Periodic Table for copper on the left: 29 protons, 35 neutrons and 29 electrons.
- Click the right arrow to see the electric charges associated with the copper atom.
- Tell students that
- each proton has one positive charge (+)
- each electron has one negative charge (–)
- electrons are often abbreviated as e– for this reason
- neutrons have no charge [A neutron walks into a bar and asks “how much for a drink?” The bartender replies “For you, no charge”]
- Since there is the same number of positively charged protons as negatively charges electrons, the net charge of the copper atom is zero.
Note: The distribution and nature of the electric subatomic particles are important. The protons hold their positive charges tightly bound with the non-charged neutrons in the nucleus. Electrons, on the other hand, are more loosely associated with the atom and in some elements may easily jump from one atom to another. This concept of electrons moving between atoms is important in the next slide as we begin to understand electric current and conductivity.
_______________________________________

SLIDE ATOM-3-4
- In elements known as conductors, electrons can be bumped from one atom to another.
- The positively charged protons in the nucleus are relatively fixed and associated with maintaining structure (of the copper wire in this case).
- As shown, an incoming electron can bump an electron from one atom to the next. The “bumped” electron, in turn, bumps an electron from another atom. Thus, in a “bucket brigade” fashion, negatively charged electrons can flow through a copper wire as shown in the illustration at the bottom of this slide.
- Electrons can move in any direction in a solid copper (or other conductor) wire. So what causes them to flow in a specific direction? The answer is shown in the next slide.
_______________________________________

SLIDE ATOM-3-5
- In an electric circuit, the battery causes the unidirectional flow of electrons by releasing electrons from its (-) terminal and attracting them to its (+) terminal.
-
- As long as the battery is “charged”, electrons will continue to flow in the direction shown. Unless the battery runs out, the wire is cut or (in this example) the light bulb is removed, electrons will continue to flow through the copper wire by jumping from one copper atom to another and bumping another electron on its way.
Note: By convention, the direction of the current is considered as moving in the opposite direction of the electron flow. In this example, the current is considered to be in a counterclockwise direction while the electron flow is in a clockwise direction. This unfortunate confusion is largely due to Benjamin Franklin, who referred to the direction of electrical currents prior to the discovery of electron flow through a circuit. This has always been a source of confusion for students ever since. We have omitted labeling the direction of the current at this time as we wish for students to focus on electrons at this point.
_______________________________________

SLIDE ATOM-3-6
-
- This slide introduces the concept of electric conductors and insulators.
- Elements with atoms that hold tightly to their electrons, not allowing them to easily be bumped off by other electrons, will not “conduct” electricity. The “bucket brigade” simply won’t get started. These elements are called insulators.
Note: Students will learn that many metals are good conductors, while nonmetals are generally not.
_______________________________________

SLIDE ATOM-3-7
- This slide introduces the Lab for this Investigation.
- As depicted in the photo, you will use multimeters to test for the ability of various elements to conduct electricity.
- Emphasize safety in the Lab. Explain to students that they will wear goggles, gloves, and lab coats for protection.
Note: To the greatest extent possible, the teacher should encourage students to visualize the subatomic events (movement of electrons between atoms) as they perform these analyses in the Lab and in discussing their results.

