Teacher Portal:
Atomic Structure
Investigation 4 – PostLab

ZERO-IN
Italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included to support the teacher’s understanding of the content being introduced within the CELL.
ANALYZE IT
Instruct students to complete the Analysis Questions in their SDRs then discuss them as a class. Use the suggested responses below to guide students’ answers.
- How did each element react when they were added to the hydrochloric acid (HCl)? Zinc (Zn) had a strong reaction to the acid (it produced many bubbles and changed color), while carbon (C) had no reaction to the acid. Silicon’s (Si) reaction was the same as the nonmetal, carbon (C).
- How effective was each element at conducting electricity? Zinc (Zn) was able to conduct electricity effectively while carbon (C) was not able to conduct electricity at all. Silicon (Si) was also able to conduct electricity; however, it was not effective at conducting electricity as zinc (Zn).
- Create a Venn Diagram in the space below using the following elements: zinc, copper, magnesium, iron, aluminum, silicon, sulfur, and carbon.
Note: Students should reflect on what they have learned about the Periodic Table of the Elements as they decide where to place the elements in the diagram. A sample student answer is provided below.
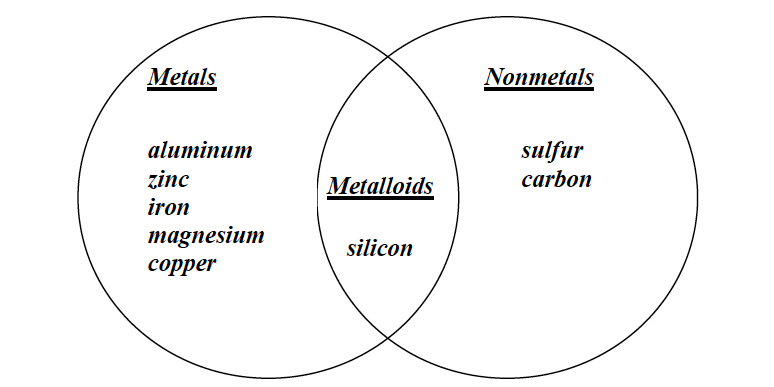
Venn diagram as a reflection of the Periodic Table
- Discuss how students decided to group the elements in the Venn diagram.
- Ask students to compare the Venn diagram that they created in Investigation 1 to the Venn diagram they completed in Investigation 4.
Note: In Investigation 1, most students probably did not have any elements that fit into both categories, whereas in Investigation 4 they should have placed silicon in the center area.

- Ask students to compare similarities between the new diagram and the Periodic Table of the Elements.
- Help students realize that their Venn diagram separated elements into metals and nonmetals similar to the way that the Periodic Table does.
- Help students realize that there is a place in both their diagram and the Periodic Table where elements can have properties of metals and nonmetals. Explain to students that the elements located between the metals and the nonmetals are called the metalloids.
GET FOCUSED
Instruct students to complete the Focus Questions in their SDRs then discuss them as a class. Use the suggested responses below to guide students’ answers.
- What is a metalloid? A metalloid is an element that can have the properties of both metals and nonmetals.
- Where are metalloids found on the Periodic Table of the Elements? Metalloids can be found on the border of the metals and nonmetals of the Periodic Table of the Elements. The metalloids are boron (B), silicon (Si), arsenic (As), tellurium (Te), germanium (Ge), antimony (Sb), polonium (Po) and astatine (At).
- How is the Periodic Table of the Elements arranged? The Periodic Table separates elements into metals, nonmetals, and metalloids. All of the elements on the left side of the border that run through Groups 13-17 are metals with the exception of hydrogen (H). The elements on the right side of the Periodic Table are nonmetals. Finally, the elements between the nonmetals and the metals are the metalloid elements.
COMPREHENSION CHECK
The Comprehension Check is designed to summarize the Core Experience Learning Lab and provide the teacher with an informal way to assess students’ understanding of the big ideas.
If time permits, ask your students to answer each Focus Question below. Use the suggested responses below to guide students’ answers.
- What is an element? Elements are substances that cannot be broken down into smaller components through chemical reactions.
- What is the Periodic Table of the Elements? The Periodic Table of the Elements is a table that organizes elements according to their physical and chemical properties.
- How is the Periodic Table of the Elements arranged? Elements with similar physical and chemical properties are grouped together on the table. For example, elements with metallic properties are grouped on the left side of the table while elements with nonmetallic properties are grouped on the right side of the table. The columns of the Periodic Table are referred to as groups and the rows are referred to as periods. The elements increase in atomic number from left to right. In Investigation Four, the Periodic Table separates elements into metals, nonmetals, and metalloids. All of the elements on the left side of the border that run through Groups 13-17 are metals with the exception of hydrogen (H). The elements in the right side of the Periodic Table are nonmetals. Finally, the elements between the nonmetals and the metals are the metalloid elements.
- How do the elements from the metal and nonmetal groups differ? Metals and nonmetals have different chemical properties from each other. For example, they react differently when combined with other substances. In our example, when we added metals to hydrochloric acid (HCl) the metals generated bubbles and heat as they reacted. Alternatively, when nonmetals were added to hydrochloric acid (HCl), no visible chemical reaction took place. In Investigation Three, Elements from the metal and nonmetal groups have different physical and chemical properties. One difference in physical properties is the ability to conduct or transmit electricity. Elements that are metals have the ability to conduct electricity while elements that are nonmetals do not.
- What are atoms? What components make up an atom? An atom is the smallest particle of an element that still has all the properties of that element. Atoms are composed of three different types of subatomic particles. In the nucleus of the atom are the protons and the neutrons. Protons have a positive electrical charge while the neutrons have no electrical charge. Orbiting the nucleus are the atom’s electrons. They are small subatomic particles that carry a negative electrical charge.
- What information about an element can be determined by using the Periodic Table of the Elements? An element’s atomic mass is equivalent to the number of protons in its atoms’ nucleus and the number of electrons normally orbiting the nucleus. In addition, the number of neutrons likely to be found in its nucleus is equal to the element’s atomic mass minus its atomic number.
- What is a metalloid? A metalloid is an element that can have the properties of both metals and nonmetals.
- Where are metalloids found on the Periodic Table of the Elements? Metalloids can be found on the border of the metals and nonmetals of the Periodic Table of the Elements. The metalloids are boron (B), silicon (Si), arsenic (As), tellurium (Te), germanium (Ge), antimony (Sb), polonium (Po) and astatine (At).
