Teacher Portal:
Atomic Structure
Investigation 4 – Lab

ZERO-IN
Italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included to support the teacher’s understanding of the content being introduced within the CELL.
MINDSET
This Investigation is designed to
- introduce students to a new type of category on the Periodic Table: metalloids,
- allow students to compare conductivity and reactivity of metalloids to metals and nonmetals through experimentation, and
- help students locate and understand the placement of the metalloids on the Periodic Table.
BE PREPARED
Teacher Preparation for the Investigation includes the following. This preparation should be done prior to students arriving in the lab.
- Fill five 100 ml beakers, one for each group, with 60 ml of 3N hydrochloric acid (HCl).
- Label a weigh dish “carbon” using masking tape and a lab marker. Prepare 10 pieces of carbon using a lab scoop. The pieces should be about 1 cm3.
- Label a weigh dish “silicon” using masking tape and a lab marker. Prepare 10 silicon lumps, 2 for each group. Each lump should be about 1 cm3 or 5 g.
- Label a weigh dish “zinc” using masking tape and a lab marker. Prepare 10 pieces of mossy zinc, 2 for each group. Each piece should be about 2 g.
- Put all materials at the distribution center.
- Divide the students into five cooperative groups.
Note: Each student lab group will need the materials listed below.
Student Preparation for the Investigation includes having students gather the following materials. This preparation takes place on lab day after student groups have settled at their assigned lab tables.
Note: The materials are listed in students’ SDRs. They are also listed below for your reference.
- (2) pieces of carbon
- (2) silicon lumps
- (2) pieces of mossy zinc
- (3) weigh dishes
- (3) 100 ml beakers empty
- (1) 100ml beaker containing 60 ml of 3N hydrochloric acid
- (1) 100 ml graduated cylinder
- (1) multimeter
- (1) red probe
- (1) black probe
- (1) wire with alligator clips
- (1) D cell battery
- (1) battery holder
- (1) stopwatch
- (1) pair of vinyl gloves per student
- (1) pair of goggles per student
- (1) Periodic Table per student
Direct one student from each lab group to collect the materials listed in their SDRs.
INVESTIGATE
- Discuss with students how they have investigated differences in the physical and chemical properties of elements and how the differences relate to the placement of the elements within the Periodic Table.
- Explain to students that during this Investigation, they will explore the physical and chemical properties of metalloids and compare them to that of the metals and nonmetals that they previously explored.
- Encourage students to reflect on the PreLab video as they move through the procedural steps.
- Explain to students that during the Experiment, every procedural step is important. If one step is skipped, data can become invalid. To help students keep on track, direct them to read each step thoroughly, complete the step, then check it off (Read it – Do it – Check it off).
- Remind students to wear gloves, safety eyewear, and lab coats when handling chemicals.
- Direct students to complete procedural steps in their SDRs.
Note: The procedural steps are listed below for your reference. Teacher “Notes” are inserted, as needed, to help facilitate the lab.
- Use the masking tape and lab marker to label three weigh dishes with the following element names: Carbon, Zinc, and Silicon.
Trial 1:
- In Trial 1, you will analyze the reactivity of silicon (Si), zinc (Zn) and carbon (C) in hydrochloric acid (HCl).
- Use a 100 ml graduated cylinder to add 20 ml of hydrochloric acid to a 100 ml beaker.
- Add 1 lump of mossy zinc to the beaker.
- Watch the beaker for 60 seconds. Use the stopwatch to keep time.
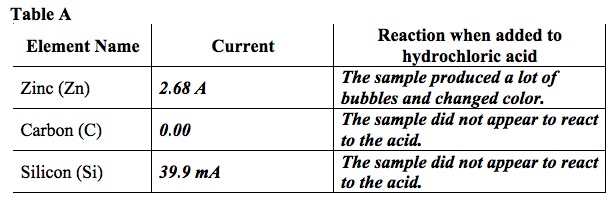
- Record any reactions in Table A.
- Use a 100 ml graduated cylinder to add 20 ml of hydrochloric acid to a second 100 ml beaker.
- Add 1 piece of carbon to the beaker.
- Watch the beaker for 60 seconds. Use the stopwatch to keep time.
- Record any reactions in Table A.
- Use a 100 ml graduated cylinder to add 20 ml of hydrochloric acid to a 100 ml beaker.
- Add 1 lump of silicon to the beaker.
- Watch the beaker for 60 seconds. Use the stopwatch to keep time.
- Record any reactions in Table A.
Trial 2:
- Prepare the multimeter. If you need additional help you can refer to the Multimeter Use and Operation in the Procedure Toolbox.

- Place the connector of a red probe into the outlet marked “10ADC” on the front of the multimeter.
- Place the connector of the black probe into the outlet marked “COM” on the front of the multimeter.
- Turn the selection dial on the multimeter to the 10A mark in the DCA range. This sets the multimeter to measure amps.
- Insert the tip of the red probe on the positive side of the battery holder between the copper strip and the plastic casing.
- Attach one end of an alligator clip wire to the tab at the negative terminal of the battery holder.
- Test the amps of the current by placing the tip of the black probe against the tip of the free end of the alligator clip.
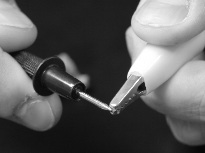
- How many amps strong is the current? 2.68 A
Note: If the current is less than 0.2 A, the multimeter should be adjusted to read in the 200 mA (milliamp) range. Adjust the multimeter as follows:
- Turn the selection dial to the “Off “ position.
- Remove the connector of the red probe from the 10ADC outlet andinto the VΩmA outlet.
- Turn the selection dial to the 200 mA mark in the DCA range and repeat steps 4 through 6. Record the current in units of mAmps (mA).
- Test electrical current as it flows through zinc.

- Place the tip of the black probe and the free end of the alligator clip against a piece of zinc. Make sure the tip of the probe and the alligator clip do not touch.
- Observe the current flowing through the circuit by reading the display on the multimeter.
- Record the current for the element in Table A.
- Repeat the previous step using the remaining elements.
Note: Some groups may have difficulty getting a proper reading on the multimeter when they are testing the mossy zinc. This is because the compound zinc oxide forms on the outside of zinc when it is exposed to oxygen in the air. Zinc oxide is the white residue that can be found on the outside of the mossy zinc pieces. If students are not getting a proper reading, have them rub the white residue off with the teeth of the alligator clip.
CLEAN UP
Let students know your expectations for clean up. Ask them to clean up.
