Teacher Portal:
Atomic Structure
Investigation 4 – Concept Day
ZERO-IN
The italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included supporting the teacher’s understanding of the content being introduced within the CELL.
ASK WHY
Explain to students that it is important to know about atoms because atoms are the building blocks of matter. We use our knowledge of atoms to explain the properties of matter, understand and manipulate matter and, ultimately, the world around us.
BRANCH OUT
Molecular biologists use the science of atoms and molecules to understand disease and develop new treatments. For example, they know that sickle cell anemia is caused by one stray molecule and have discovered which molecular errors cause some cancers.
PRINT IT
Use your browser to download a printable PDF as a help during the slide presentation and to make additional notes. In your browser, go to File > Print and then choose to save as PDF.
NAVIGATE IT
Once the slide presentation is launched
- use your left and right arrows to advance or go back in the slide presentation, and
- hover your mouse over the left edge of the presentation to get a view of the thumbnails for all the slides so that you can quickly move anywhere in the presentation.
- Click HERE to launch the slide presentation for the CELL.
SHARE IT

SLIDE ATOM-4-1
- In this final Investigation for Atomic Structure, we will return to the Periodic Table and focus on the metalloids.
Note: As there are really no new concepts or lab procedures introduced in this Investigation, we will take the opportunity to introduce the concept of molecular weight.
__________________________________________________

SLIDE ATOM-4-2
Note: The Periodic Table of the Elements is presented in this slide to return students’ thinking to elements and atoms and the number of different kinds of information the Table provides.
- Use this opportunity to review:
- Groups
- Periods
- Atomic number
- Atomic mass
- Subatomic particles
- Etc.
__________________________________________________

SLIDE ATOM-4-3
Note: This slide is included to remind students that atoms can combine to form molecules and compounds. It is important to recall that compounds are molecules too. They are molecules that include more than one kind of element.
- In this example, O2, (oxygen gas) would be referred to as a molecule, but not a compound. Water (H2O) on the other hand, can be referred to as a compound because it contains two different atoms. However, H2O could also be referred to as a molecule. In fact, in most cases compound molecules are simply referred to as “molecules”.
__________________________________________________

SLIDE ATOM-4-4
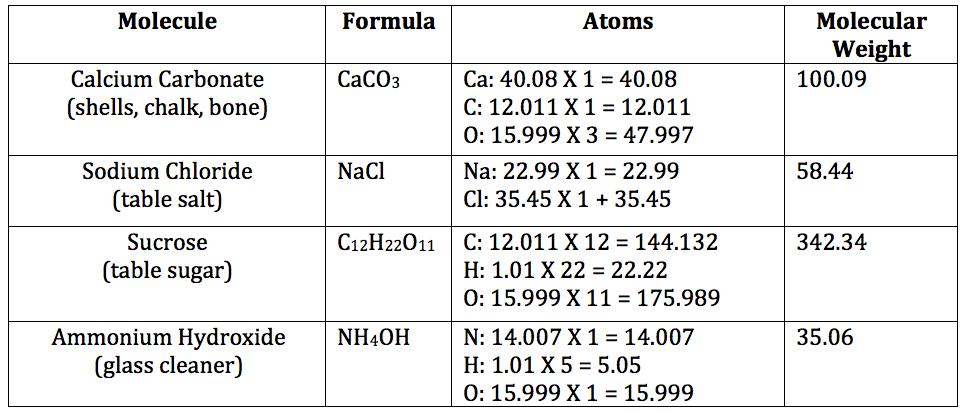
Note: Students will not actually work with molecular weight until high school when they use the concept to determine molar concentrations. However, while we are not making direct use of molecular weight at this point, it is so easily calculated given what students have already learned in Atomic Structure, that we should take a moment for an initial introduction.
Note: The molecular weight (also called molecular mass or formula weight) of a molecule is derived simply by adding the atomic masses of each of the atoms in the molecule. Atomic mass can be found for every atom on the Periodic Table of the Elements. The important thing to remember is that if more than one atom of a particular element is found in the molecule, its mass must be counted that many times.
- Read the slide to the students.
- If time permits, practice the calculations below as a class:
__________________________________________________

SLIDE ATOM-4-5
Note: This slide initiates the discussion of metalloids.
Note: ANIMATIONS are arranged so that metals, nonmetals and metalloids are given individual “clicks” in case the teacher wishes to discuss them individually as they are circled on the Periodic Table.
__________________________________________________

SLIDE ATOM-4-6
Note: This simple slide lists some of the properties of metals and nonmetals. The metalloids are generally intermediate between metals and nonmetals in their properties. Metalloids are used extensively in the computer industry. They are also used as flame-retardants and in glass manufacturing.
- Read the slide to the students.
__________________________________________________

SLIDE ATOM-4-7
Note: This final slide introduces students to the Lab for this Investigation. As can be seen, it is composed of two procedures they have already performed in this Investigation, chemical reactivity, and electrical conductivity.
- Explain to students that in Lab, they will analyze the chemical reactivity and electrical conductivity of three elements: zinc ( a metal), carbon (a nonmetal), and silicon (a metalloid).
- Emphasize safety in the Lab. Explain to students that they will be handling various elements and chemicals and must wear goggles, gloves, and lab coats for protection.
Note: A safety reminder is given for handling hydrochloric acid. As is the case with many caustic acids and bases, HCl is virtually indistinguishable from water by simple visual inspection. Students must be taught to be consistently safe in the Lab.

