Teacher Portal
Acids and Bases
Investigation 3 – PreLab
ZERO-IN
Italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included to support the teacher’s understanding of the content being introduced within the CELL.
ASK WHY
Remind students that acids and bases are all around us. They play an important role in the digestion of the foods we eat, the medicine we take, and even the cleaning products we use. Understanding these two classes of compounds can help us stay safe, healthy, and be informed consumers.
BRANCH OUT
Remind students that clean water is essential for everyday life. Water treatment plant and system operators ensure that the water pumped from rivers, streams, and reservoirs is clean and has a safe pH before it gets to your home.
GET FOCUSED
Inform students that the Investigation is designed to help them to answer the following Focus Question:
- What happens to the concentration of hydrogen ions (H+) when an acid and a base are reacted? When an acid and a base react, H+ ions and OH– ions react to produce water and decrease the concentration of H+ ions.
Note: This question is located in students’ SDRs at the beginning and end of the Investigation.
Note: This is a succinct response to the Focus Question and is placed here for your reference at this time. Fully developed responses to the Focus Questions can be found on the PostLab page.
GO DEEPER
As a class, read the Background(s) in the Investigation. Have students read the information aloud or silently to themselves. When students have finished, discuss the following concepts as a class:
- The concentration of hydrogen ions (H+) in acidic solution can be decreased by diluting the solution.
- Another way to decrease the concentration of H+ ions in a solution is to perform a chemical reaction by dissolving both an acid and a base in water.
- Discuss the following equation illustrating that hydrochloric acid, like all acids, dissociates into H+ ions and a negatively charged ion like Cl– when dissolved in water.
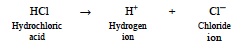
- Discuss the following equation illustrating that sodium hydroxide, like all bases, dissociates into OH– ions and a positively charged ion like Na+ when dissolved in water.

- Discuss the following equation illustrating the neutralization reaction between hydrochloric acid and sodium hydroxide to produce water and NaCl.
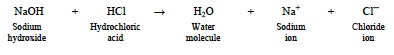
- If the acid concentration is higher than the concentration of the base, then the H+ concentration will be high and the pH will remain in the acidic range.
- If the concentrations of the acid and base are equal, the pH will be in the neutral range, approximately 7.
- If the base concentration is higher than the acid concentration, then the H+ concentration will be very low and the pH will be in the basic range.
- Discuss the following equation describing the neutralization reaction. The reaction consumes two acid molecules and produces two water molecules because unlike NaOH, Mg(OH)2 possesses two OH– ions.

Note: These concepts are integrated into the Background(s) and are used to deepen students’ comprehension of the big ideas.
LEARN THE LabLearner LINGO
The following list includes Key Terms that are introduced in the Investigation Background(s). They should be used, as appropriate, by teachers and students during everyday classroom discourse.
- neutralization
Note: The definition of this term can be found on the Introduction page to the CELL.
Note: Additional words may be bolded within the Background(s). These words are not Key Terms and are strictly emphasized for exposure at this time.
SET FOR SUCCESS
- Direct students to complete the Recall section in their SDRs. Student answers will vary.
- What is the relationship between the pH of a solution and the concentration of hydrogen ions (H+)?
- When the pH is low, is the concentration of hydrogen ions (H+) low or high?
- When the pH is high, is the concentration of hydrogen ions (H+) low or high?
- How can the concentration of hydrogen ions (H+) be changed?
- Inform students that the Investigation will help them to understand what happens to the concentration of hydrogen ions (H+) when an acid and base react.
- Play the video below. Stop to ask students questions or answer students’ questions when necessary. Remind students to follow along with their SDRs and make any notes that they think might be helpful.
- After the video, direct students to divide into their lab groups to discuss their strategy for the lab. For example, they may assign certain group members to perform specific functions during the lab.
Note: The purpose of the video is to allow students to anticipate the laboratory experience they will soon encounter. Students should leave this PreLab session with a firm idea of what to expect and how to perform in the lab.
Note: Homework is posted below the video.
HOMEWORK
Tell students that they should review the Investigation in preparation for the Lab.

