Teacher Portal:
Acids and Bases
Investigation 3 – PostLab

ZERO-IN
Italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included to support the teacher’s understanding of the content being introduced within the CELL.
ANALYZE IT
Instruct students to complete the Analysis Questions in their SDRs then discuss them as a class. Use the suggested responses below to guide students’ answers.
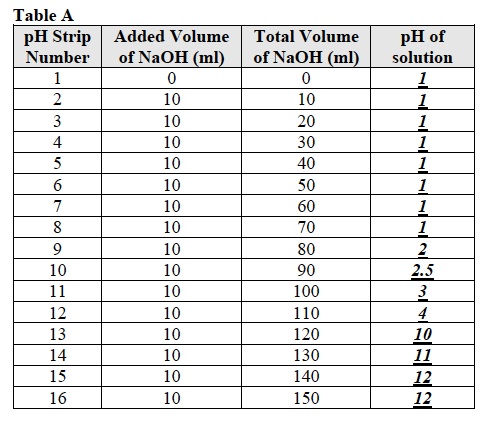
Note: Students will begin the Analysis by creating a graph that compares the pH of the solutions and the total volume of sodium hydroxide (NaOH) that they added to the hydrochloric acid (HCl) solution.
Note: Students should use the data from Table A to draw the graph.
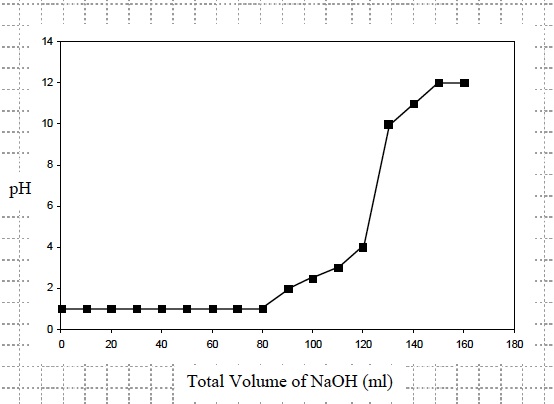
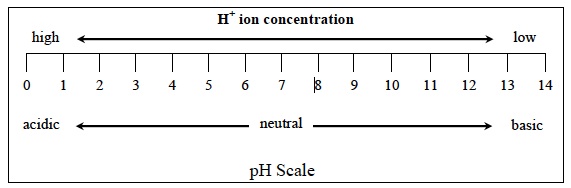
- What was the pH of the hydrochloric acid (HCl) solution before any sodium hydroxide (NaOH) was added? Was it in the acidic, neutral, or basic region of the pH scale? The pH of the solution was 1 which is in the acidic region.
- What happened to the pH of the hydrochloric acid (HCl) solution after you added between 10 and 80 ml of sodium hydroxide (NaOH)? Is this in the acidic, neutral, or basic region of the pH scale? The pH of the solution remained at pH 1which is in the acidic region.
- What happened to the pH of the hydrochloric acid (HCl) solution after you added between 80 and 110 ml of sodium hydroxide (NaOH)? Is this in the acidic, neutral, or basic region of the pH scale? The pH of the HCl solution increased from 1 to 3 which is in the acidic region.
- What happened to the pH of the hydrochloric acid (HCl) solution after you added between 110 ml and 130 of sodium hydroxide (NaOH)? Is this in the acidic, neutral, or basic region of the pH scale? The pH of the solution increased from 3 to 10 which is in the basic region.
- What happened to the pH of the hydrochloric acid (HCl) solution after you added between 130 ml and 160 of sodium hydroxide (NaOH)? Is this in the acidic, neutral, or basic region of the pH scale? The pH of the solution increased from 10 to 12 which is in the basic region.
- What happened to the concentration of hydrogen ions (H+) ions when the pH increased from 1 to 12? The concentration of H+ ions decreased when the pH increased from 1 to 12.
- Look at the graph. What total volume of sodium hydroxide (NaOH) would need to be added to make the solution neutral (pH 7)? Approximately 125ml of NaOH would need to be added to make the pH of the solution 7.
- Compare the concentration of H+ ions in the acidic, neutral, and basic regions of your graph. Which region has the higher, lower, and intermediate concentrations of H+ ions? The acidic region has the higher concentration, the basic region has the lower concentration and the neutral region has the intermediate concentration of H+ ions.
GET FOCUSED
Instruct students to complete the Focus Question in their SDRs then discuss it a class. Use the suggested response below to guide students’ answers.
- What happens to the concentration of hydrogen ions (H+) when an acid and a base are reacted? When an acid and a base react, H+ ions and OH– ions react to produce water and decrease the concentration of H+ ions.
COMPREHENSION CHECK
The Comprehension Check is designed to summarize the CELL and provide the teacher with an informal way to assess students’ understanding of the big ideas.
If time permits, ask your students to answer each question below. Use the suggested responses below to guide students’ answers.
- What properties make acids and bases chemically reactive? Acids release H+ ions when they are mixed with water and bases remove H+ ions when they are mixed with water.
- What is the relationship between the concentration of H+ ions in a solution and the chemical reactivity of strong and weak acids? Stronger acids are able to release more H+ ions when mixed with water resulting in a higher concentration of H+ ions. Weaker acids release fewer H+ ions resulting in a lower concentration of H+ ions.
- What is the relationship between the concentration of H+ions in a solution and the chemical reactivity of strong and weak bases? Stronger bases are able to remove more H+ ions when mixed with water resulting in a lower concentration of H+ ions. Weaker bases remove fewer H+ ions resulting in a higher concentration of H+ ions.
- What is the relationship between the pH of a solution and the concentration of hydrogen ions (H+)? As the pH of a solution increases, the concentration of H+ ions decreases. An increase of 1 pH unit decreases the concentration 10 times. As the pH of a solution decreases, the concentration of H+ ions increases. A decrease of 1 pH unit increases the concentration 10 times.
- What happens to the concentration of hydrogen ions (H+) when an acid and a base are reacted? When an acid and a base react, H+ ions and OH– ions react to produce water and decrease the concentration of H+ ions.
