Teacher Portal:
Acids and Bases
Investigation 2 – PostLab

ZERO-IN
Italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included to support the teacher’s understanding of the content being introduced within the CELL.
ANALYZE IT
Instruct students to complete the Analysis Questions in their SDRs then discuss them as a class. Use the suggested responses below to guide students’ answers.
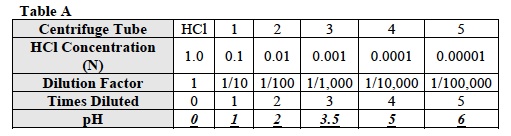
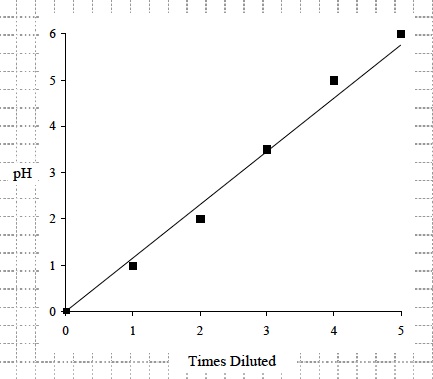
- Draw a graph that compares the pH of the solutions and the number of times you diluted the original solution of HCl. Use the data from Table A to draw the graph.
- What solvent did you use to dilute your solutions as you performed your serial dilutions? Water
- Look at the solutions in the centrifuge tubes. As you diluted the original red solution of hydrochloric acid (HCl), what happened to the red color of the hydrochloric acid (HCl)? Which solutions displayed a shade of red or pink and which solutions were colorless? The red color became fainter until the solutions were colorless. Solutions 1 and 2 displayed some shade of red or pink but solutions 3, 4, and 5 were colorless.
- Based on your observations of the color of the solutions, what happened to the concentration of the red food coloring when you diluted the original red solution of hydrochloric acid (HCl)? The concentration of the food coloring was decreased by the dilution until it was so low that it could not be detected.
- What other solutes in addition to the food coloring were you diluting and measuring as you performed your serial dilutions? Hydrochloric acid (HCl)
- From the data in Table A, what was the pH of the undiluted HCl? The pH of the undiluted HCl was 0. 7.
- Look at your graph. As the undiluted hydrochloric acid (HCl) was diluted from 1 to 5 times with water, what happened to the pH? The pH increased each time the hydrochloric acid (HCl) was diluted.
- Inspect your graph. As the original hydrochloric acid (HCl) was diluted from 1 to 5 times with water, what happened to the hydrogen ion (H+) concentration? The hydrogen ion (H+) concentration decreased each time the HCl was diluted.
- Look at Table A. Solution 1 was how many times less concentrated than the hydrochloric acid (HCl)? How could you use a decimal to show this change? Solution 1 was 10 times less concentrated than the hydrochloric acid (HCl). The decimal, 0.1 can be used to show this change.
- Look at Table A. Solution 2 was how many times less concentrated than the original solution of HCl? How could you use a decimal to show this change? Solution 2 was 10 times less concentrated than solution 1. The decimal, 0.01can be used to show this change.
- Each solution was how many times less concentrated than the previous one? 10 times
GET FOCUSED
Instruct students to complete the Focus Question in their SDRs then discuss it as a class. Use the suggested response below to guide students’ answers.
- What is the relationship between the pH of a solution and the concentration of hydrogen ions (H+)? As the pH of a solution increases, the concentration of H+ ions decreases. An increase of 1 pH unit decreases the concentration 10 times. As the pH of a solution decreases, the concentration of H+ ions increases. A decrease of 1 pH unit increases the concentration 10 times.
