Teacher Portal:
Acids and Bases
Investigation 1 – Concept Day
ZERO-IN
Italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included to support the teacher’s understanding of the content being introduced within the CELL.
ASK WHY
Explain to students that acids and bases are all around us. They play an important role in the digestion of the foods we eat, the medicine we take, and even the cleaning products we use. Understanding these two classes of compounds can help us stay safe, healthy, and be informed consumers.
BRANCH OUT
Explain to students that hematologists monitor the delicate balance between acids and bases in the blood. They know that if the acidity of the blood changes too much, the blood loses its capacity to carry oxygen, and, without oxygen, cells die.
PRINT IT
Use your browser to download a printable PDF as a help during the slide presentation and to make additional notes. In your browser, go to File > Print and then choose to save as PDF.
NAVIGATE IT
Once the slide presentation is launched
- use your left and right arrows to advance or go back in the slide presentation, and
- hover your mouse over the left edge of the presentation to get a view of the thumbnails for all the slides so that you can quickly move anywhere in the presentation.
- Click HERE to launch the slide presentation for the CELL.
SHARE IT

SLIDE ACIDS-1-1
Note: In this Investigation, we wish to introduce the concept of pH. We wish to emphasize that pH is a chemical property of matter. pH involves both acids and bases containing an excess of either H+ or OH– ions, respectively. Finally, we will show that various solutions that students commonly come in contact with each have their own characteristic pH. As a result, students will see that solutions of varying pHs are very useful in our lives.
______________________________________________

SLIDE ACIDS-1-2
- This slide introduces the concept of pH.
- pH is a chemical property as opposed to a physical property of matter.
- Read and discuss the slide with the students.
Note: A brief review of physical and chemical properties may be useful at this juncture. The following table lists a few physical and chemical properties of matter:
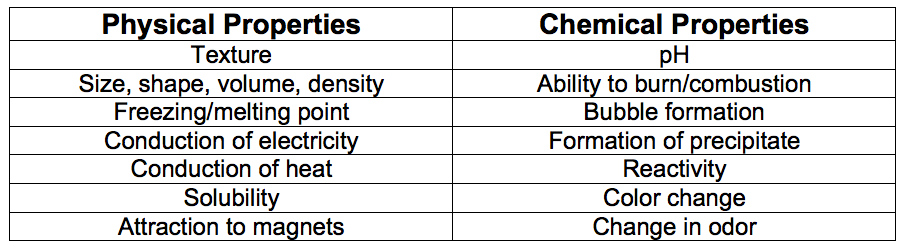
______________________________________________

SLIDE ACIDS-1-3
- This slide develops the concept that an acid contains high concentrations of hydrogen ions (H+).
- An ion is a chemical compound that has a positive or negative charge.
- A common acid, and one we use often in LabLearner, is hydrochloric acid (HCl). The slide shows how HCl dissociates into a negative (Cl–) and positive (H+) ion in water. It is the presence of H+ that gives the solution its acidic properties, not the presence of the chloride ion (Cl–).
- The hydrogen ion readily interacts with other molecules and compounds and gives acids their major chemical property of being very reactive.
______________________________________________

SLIDE ACIDS-1-4
- This slide states that “The greater the concentration of H+ ions, the lower the pH and the more acidic (less basic)” , thus introducing the relationship between H+ and the pH scale.
- The graphic in this slide helps us visualize that many more hydrogen ions are present in solutions of low pH than at higher pH values.
- Stronger acids are found at the lowest pHs.
______________________________________________

SLIDE ACIDS-1-5
- This slide develops the concept that a base contains high concentrations of hydroxide ions (OH–).
- A common base, and one we use often in LabLearner, is Sodium Hydroxide (NaOH).
- The slide shows how NaOH dissociates into a negative (OH–) and positive (Na+) ion in water.
- It is the presence of OH– that gives the solution its basic properties, not the presence of the sodium ion (Na+).
______________________________________________

SLIDE ACIDS-1-6
- This slide states that “The greater the concentration of OH– ions, the higher the pH and the more basic (less acidic)”, thus introducing the relationship between OH– and the pH scale.
- The graphic in this slide helps us visualize that many more hydroxide ions are present in solutions of high pH than at lower pH values.
- Stronger bases are found at the highest pHs.
______________________________________________

SLIDE ACIDS-1-7
- This slide pulls together the concepts from the previous four slides.
- The graphic at the top integrates the notion of low pHs having high H+ concentrations and high pHs having high OH– concentrations.
Note: Looking at the pH scale in this slide you may notice that at a pH of 7 (neutral pH), there are a few H+ ions and a few OH– ions. The solution at this point would be neutral because the OH– and H+ ions would interact with each other and form the molecule water (H2O). Thus neither the H+ ion nor the OH– ion would be present in greater amounts than the other. Consequently, even though there may be some H+ ions at very high pH, the vast excess of hydroxide ions would bind them up and the net result would be elevated OH– ions. The reverse would hold true at the opposite end of the pH scale with hydrogen ion vastly exceeding free hydroxide ions. DEPENDING on the class, this discussion may become confusing and could be omitted simply by accentuating the high concentrations of H+ at low pH and high concentrations of OH– at high pHs.
- Use the graphic table at the bottom of this slide as an easy reference and review of pH.
______________________________________________

SLIDE ACIDS-1-7
- This slide serves as an introduction for the Investigation 1 Lab. Each of the compounds that will be tested in the lab is listed along with their chemical names and chemical formulae.
______________________________________

SLIDE ACIDS-1-8
Note: Students have seen this slide previously in earlier CELLs.
- This slide is shown as a reminder to wear gloves, goggles, and lab coats when working with strong acids and bases.
Note: It is also instructive as an illustration of just how reactive both the hydrogen and hydroxide ion can be. The skin contains many different kinds of molecules that undergo chemical reactions when exposed to H+ or OH–. Evidence of a chemical reaction, such as bubble formation, heat production, and color and odor change all accompany exposure of skin tissue to strong acids or bases. Careful!

