Teacher Portal:
Chemical Reactions
Investigation 3 – Concept Day
ZERO-IN
Italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included to support the teacher’s understanding of the content being introduced within the CELL.
ASK WHY
Remind students that everything in the environment, whether naturally occurring or man-made, is composed of chemicals, and research of chemicals has led to the discovery and development of new and improved paints, medicines, cosmetics, electronic components, and thousands of other products.
BRANCH OUT
Explain to students that pharmacists distribute prescription drugs to people. To do this safely, they must understand the chemical composition of these drugs so that they can create special versions of medications for patients who may have allergies to certain substances, give the right dosage, and give patients advice on the possible side effects.
PRINT IT
Use your browser to download a printable PDF as a help during the slide presentation and to make additional notes. In your browser, go to File > Print and then choose to save as PDF.
NAVIGATE IT
Once the slide presentation is launched
- use your left and right arrows to advance or go back in the slide presentation, and
- hover your mouse over the left edge of the presentation to get a view of the thumbnails for all the slides so that you can quickly move anywhere in the presentation.
- Click HERE to launch the slide presentation for the CELL.
SHARE IT

SLIDE CHEM-3-1
- Remind students that during Investigation 1 they followed the chemical reaction by measuring heat as a product, and in Investigation 2 they followed the chemical reaction by measuring the production of hydrogen gas.
- Inform students that during this final Investigation, they will follow one more reaction, but this time they will measure the disappearance/consumption of one of the reactants (iodine).
- Inform students that they will also discuss the reaction rate, thus introducing the element of time into the study of chemical reactions.
______________________________________________

SLIDE CHEM-3-2
- Remind students that this slide was used earlier in this Investigation in a discussion of Conservation of Matter.
- Tell students that it is used here again, this time to point out that as products of a reaction are formed, the reactants must be consumed.
- Explain that before the chemical reaction begins (for left), there are no products, only reactants. When the chemical reaction is complete (far right), there are no reactants, only products. However, during the reaction, there is a mixture of products and reactants.
- Explain that the conversion of reactants to products is a process that takes place over time. Therefore, if we were to look at the reactants and products at different time points during the reaction, we would find a mixture of the reactants and products.
- Explain that the conversion of reactants to products allows chemists to follow the rate of a chemical reaction by observing either the formation of a product or the disappearance/consumption of one of the reactants.
Note: In this Investigation, students will measure the consumption of one of the reactants, iodine (I2) as discussed further in the next slide.
______________________________________________

SLIDE CHEM-3-3
- Inform students that this slide introduces the chemical reaction that will be studied in Lab. In this reaction, acetone and iodine react to yield monoiodoacetone and hydrogen iodide.
Note: A very important new point introduced with this chemical reaction is the catalyst, in this case, hydrochloric acid.
- Explain that a catalyst increases the rate of a chemical reaction without being consumed in the reaction.
Note: In biological systems, enzymes often serve as catalysts for biochemical reactions. Students will come in contact with catalysts again and again as they advance in science.
- Emphasize that catalysts are not consumed at all in the reaction they catalyze. In fact, there will be the exact same amount of the catalyst at the end of the reaction as at the beginning.
- Explain that catalysts are not included in either the reactant or product side of the chemical equation.
- Explain that catalysts are typically written over the arrow in chemical equations.
- Read the equation to the students. “The reaction between acetone and iodine, in the presence of the catalyst hydrochloric acid, yields monoiodoacetone and hydrogen iodide.”
Note: Our atomic accounting would be as shown below: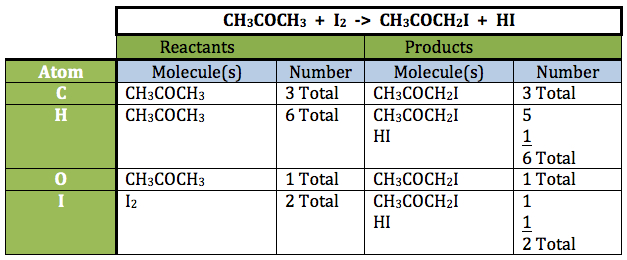
- Emphasize that a catalyst in one chemical reaction may be either a reactant or product of different chemical reactions.
Note: Students should not be allowed to get confused seeing hydrochloric acid functioning as a catalyst in this Investigation when it was one of the reactants in the reactions they studied in the previous two Investigations.
- Inform students that they will follow the reaction by observing the consumption of one of the reactants (iodine, I2) in Lab.
Note: This is again different from the previous two Investigations in which the chemical reactions were followed by measuring some aspect of product formation. Taken together, the three rather simple and safe reactions in this CELL give students exposure to a fairly wide range of chemical reactions.
______________________________________________

SLIDE CHEM-3-4
- Inform students that this slide introduces the use of light absorption and the spectrophotometer as a means of following a chemical reaction.
- Explain that any chemical reaction in which any of the reactant or product molecules has a color that is missing on the other side of the equation, permits the use of light absorption to follow it.
- Explain that one either follows the disappearance of the color (if the color-absorbing molecule is on the reactant side) or the appearance of the color (if the color-absorbing molecule is on the reactant side).
Note: This slide also has an insert that shows an absorption spectrum for iodine. [Disregard the part of the tracings to the far left as they are in the UV, not visible, range.] In the visible range, the absorption spectrum for iodine is from about 400nm to 700nm (nm stands for nanometers, the wavelength).
- Explain that iodine absorbs best at about 460nm. Students will therefore set their spectrophotometers at a wavelength of 460nm to follow iodine consumption in Lab.
Note: Most of the content for this Investigation has been covered at this point. However, the next few slides are devoted to trying to avoid or relieve a point of confusion in the minds of many students regarding the color one observes of an object and the color that that object absorbs.
______________________________________________

SLIDE CHEM-3-5
- Inform students that the background for this slide is the visible spectrum with approximate wavelengths (in nanometers) at the top.
- Explain that an apple and some blueberries are positioned on the spectrum in the region where they absorb light. That is to say, the apple and blueberries contain certain molecules that absorb light waves in these regions.
Note: Students sometimes find this counterintuitive. It seems as though a red apple must absorb red wavelengths and that a blueberry must absorb blue wavelengths of light. However, the fact is that if red light was absorbed by an apple, light energy in that range and color would not bounce off the apple’s surface and enter our eye! The same situation exists for the blueberries. If blueberries absorbed light in the blue region of the visible spectrum, light energy in that range and color would not bounce off the blueberries’ surface so we could perceive it.
______________________________________________

SLIDE CHEM-3-6
- Explain that the colored light absorbed by an object is the very color we cannot see.
- Emphasize that the light that an object does not absorb, it reflects. It is the reflected light that enters our eyes and activated cells in our retina. We perceive the reflected color.
- Explain that an apple looks red because it is composed of molecules that absorb most other wavelengths of light but reflects red light. A blueberry is blue because it absorbs most wavelengths of light but reflects blue light.
- Explain that iodine is reddish brown because it absorbs most wavelengths of light but reflects reddish-brown light.
- Tell students that if we want to measure how much iodine is left as a reactant in a chemical reaction by the amount of light it absorbs, we need to measure at a wavelength that iodine absorbs, not reflects. The absorption peak for iodine is about 460nm. Consequently, we set our spectrophotometers at 460nm.
______________________________________________

SLIDE CHEM-3-7
Note: This is a simple and fun slide to drive the light absorption concept home again with one final, very recognizable example.
- Tell students that the ocean is blue because water absorbs colors in the red part of the light spectrum.
- Explain that if ocean water absorbed colors in the blue part of the light spectrum, it would look less the color of blueberries and more the color of apples. Our planet would certainly look quite different.

Red Earth. Photo of the Earth from space in which blue pigments where replaced with red. This is an approximation of what the Earth would look like if water absorbed in the blue wavelength range instead of the red!
______________________________________________

SLIDE CHEM-3-8
- Remind students that caution must be taken when working with caustic chemicals such as hydrochloric acid and flammable chemicals such as acetone.
- Explain that lab coats, goggles and gloves should be worn throughout this Investigation.

