Teacher Portal:
Chemical Reactions
Investigation 1 – Concept Day
ZERO-IN
Italicized font represents information to be shared orally or physically completed with the students at this time.
The non-italicized font represents additional information included to support the teacher’s understanding of the content being introduced within the CELL.
ASK WHY
Explain to students that everything in the environment, whether naturally occurring or man-made, is composed of chemicals, and research of chemicals has led to the discovery and development of new and improved paints, medicines, cosmetics, electronic components, and thousands of other products.
BRANCH OUT
Explain to students that chemical engineers design and develop chemically based products such as rubber, plastic, soaps, and gasoline. They also test these products to make sure they are safe for consumers and the environment.
PRINT IT
Use your browser to download a printable PDF as a help during the slide presentation and to make additional notes. In your browser, go to File > Print and then choose to save as PDF.
NAVIGATE IT
Once the slide presentation is launched
- use your left and right arrows to advance or go back in the slide presentation, and
- hover your mouse over the left edge of the presentation to get a view of the thumbnails for all the slides so that you can quickly move anywhere in the presentation.
- Click HERE to launch the slide presentation for the CELL.
SHARE IT

SLIDE CHEM-1-1
- Inform students that this Investigation is designed to help them understand that there are common features among chemical reactions.
Note: In Investigations 1 through 3 we will study three different reactions. In this Investigation, we will follow the reaction:
NH4OH + HCl → H2O + NH4Cl
Note: This is an exothermic reaction and therefore gives off heat. Students will measure the heat released by the reaction in a calorimeter to watch the reaction progress. They will increase the amount of reactants in three Trials and see what effect reactant concentration has on product formation.
______________________________________________

SLIDE CHEM-1-2
- Inform students that this slide introduces the important concept of reactants and products.
- Read the slide to the students.
- Explain that the reactant molecules are altered by the chemical reaction and reconfigured into the product molecules. This is depicted by the different shapes and colors of the reactant molecules and the product molecules.
Note: It is important for students to understand that the atoms in the reactant molecules rearrange themselves into the product molecules. This will be more apparent on the next slide.
_____________________________________________

SLIDE CHEM-1-3
- Inform students that this slide presents two chemical reactions that introduce real reactants and products instead of the colored symbols on the previous slide.
- Explain to students that they can “read” a chemical equation as a sentence.
- Read the slide to the students.
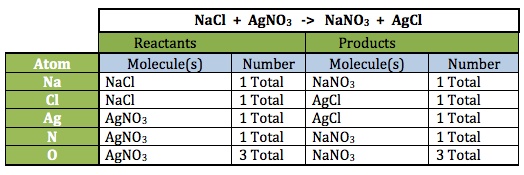
Note: The first equation is read, “The reaction between sodium chloride and silver nitrate (the reactants) yields (the arrow) sodium nitrate and silver chloride (the products).
- Tell students that all chemical equations may be read like this.
- Emphasize that every atom in the reactants turns up in the products in the exact same amount. Atoms in the reactants and the products MUST be the same; otherwise, matter would be created or destroyed in the chemical reaction!
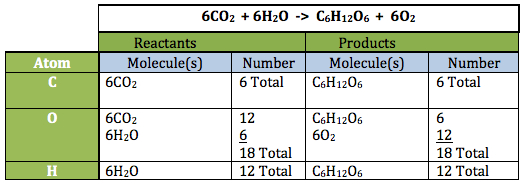
Note: The tables below provide a “bookkeeping” strategy for being sure all atoms on both sides (reactants and products) are accounted for. Notice in the second reaction (this is the reaction for photosynthesis) one must take into account the number of molecules of each reactant and product:
- Explain that the conservation of atoms in the reactants and products leads to one of the most fundamental principles of physics and chemistry, the Conservation of Matter.
Note: The Conservation of Matter is further discussed in the next slide.
_____________________________________________

SLIDE CHEM-1-4
- Inform students that this slide presents a simple demonstration to graphically illustrate Conservation of Matter.
- Explain that when the valve is opened on the upper reservoir, the blue liquid flows into the flask below. At any point in the process, the fluid is either in the reservoir or the flask. But the total amount of the fluid will always be exactly the amount that was present at the beginning.
- Emphasize that no additional fluid is introduced in the process. In the end, all of the fluid that was in the reservoir ends up in the flask.
- Explain that this is analogous to the situation of any given atom in a chemical reaction. The exact amount of atoms in the reactants must also be present in the reactants – no less and no more.
- Emphasize that if more atoms were to be present in the products than in the reactants, the matter would have been created. If fewer atoms were present in the products than in the reactants, the matter would have been destroyed.
- Explain that the Law of Conservation of Matter tells us that neither of these two things can happen.
_______________________________________________

SLIDE CHEM-1-5
Note: This slide is used to refresh students’ memories of several types of evidence that a chemical reaction has occurred. They will have seen this slide in previous CELLs.
- Remind students of the three forms of evidence that a chemical reaction has occurred.
- Explain that all three forms will be used in the three Investigations of this CELL.
Note: In this Investigation, students will follow a chemical reaction by the heat it releases. Gas formation and color change will be studied in Investigation 2 and Investigation 3, respectively.
_______________________________________________

SLIDE CHEM-1-6
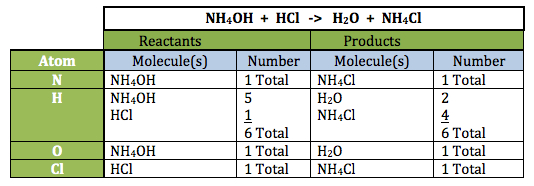
- Inform students that this slide presents the reaction they will perform in Lab. The equation for the reaction is as follows:
NH4OH + HCl -> H2O + NH4Cl
- Explain that this is an exothermic reaction. That is, it releases heat as the reaction takes place.
- Tell students that they will measure the amount of heat released from the reaction using a simple Styrofoam calorimeter.
Note: Calorimeters can be much more sophisticated and accurate than the simple one students will make and use in this Investigation. Note: This slide is set up with a series of three mouse click “animations” for you to walk students through. Click 1:
- Explain that the red line depicts the addition of ammonium hydroxide (a reactant) to the calorimeter.
Click 2:
- Explain that the second red line depicts the addition of hydrochloric acid (a reactant) to the calorimeter.
Click 3:
- Explain that students should follow the reaction by monitoring heat production.
The red lines for addition of each product and the line and red text for heat release are added to the slide with individual clicks. Note: As an aid, accounting of the individual atoms in the reaction is shown below:
_______________________________________________

SLIDE CHEM-1-7
- Inform students that this final slide accentuates the caution that must be taken when working with caustic chemicals such as hydrochloric acid.
- Emphasize that lab coats and gloves should be worn throughout this Investigation.
- Emphasize that students must wear their goggles to protect their eyes, not resting on the top of their heads or dangling around their necks. Just as there is no time to quickly put on a seatbelt in a car just before an accident, there is no time to quickly put on goggles when acid is splashed or another lab accident takes place! Always be safe.
_______________________________________________

